|
The U.S. announced Monday it would share its entire supply of AstraZeneca’s Covid-19 vaccine with other countries and readied an aid package for India, as President Joe Biden pivots to ramp up U.S. pandemic assistance to the rest of the world. The decision would make as many as 60 million vaccine doses available for export in coming months, if they clear a federal safety review. The plan represents a shift for Biden, who has hesitated to give vaccine doses and materials to other countries while focusing on ensuring Americans are vaccinated first. “Given the strong portfolio of vaccines that the United States has already authorized and that is available in large quantities — including two two-dose vaccines and one one-dose vaccine — and given AstraZeneca is not authorized for use in the United States, we do not need to use AstraZeneca in our fight against Covid over the next few months,” White House Press Secretary Jen Psaki told reporters on Monday.
The announcement reflects growing pressure the U.S. has faced to share its vaccine supply with other countries, such as India, that have seen surges of Covid-19 infections or struggled to jump-start their vaccination drives. It also shows that the Biden administration is confident in its vaccine stockpile, especially after sites over the weekend were cleared to resume administering the Johnson & Johnson one-dose shot. Biden in a phone call Monday pledged his full support to Indian Prime Minister Narendra Modi, who is combating the world’s largest surge of infections. The assistance package offered to India, which includes raw materials for vaccines and therapeutics, reflects Biden’s belief that the pandemic won’t end unless the U.S. offers assistance to other countries, according to a senior administration official. Sharing DosesNo final decisions have been made on which countries will receive the AstraZeneca doses, a senior administration official said. The U.S. previously loaned 4.2 million AstraZeneca shots to Mexico and Canada, the only Covid-19 vaccine doses it has shared so far. AstraZeneca has not sought Food and Drug Administration authorization for its vaccine, but has already manufactured millions of doses in the U.S. under a federal contract. About 10 million AstraZeneca doses that have already been produced could become available for export in the next several weeks if they pass FDA checks for product quality, a senior administration official said. Another 50 million doses are still in production and could be ready to ship by May or June if they pass the inspections, according to the official. Any decision to send doses to India could increase pressure from other countries that have been trying to get the U.S. to share shots but so far have been rebuffed. Mexico and Canada have asked for a larger allotment of AstraZeneca vaccines. “We’re in the planning process at this point,” Psaki said. More than 1.02 billion vaccine doses have been administered worldwide as of Monday, enough to fully vaccinate 6.7% of the global population, according to Bloomberg’s Vaccine Tracker. Still, the vaccination campaign has been uneven, with wealthier countries generally ahead of less wealthy ones in giving shots. Just 5.3% of India’s population has been fully vaccinated as of Monday compared with 36% in the U.S. The need for vaccines in India has become even more acute as it has become the global epicenter of the pandemic. The country recorded 352,991 new virus cases on Sunday, the fifth consecutive day it set a world record for a single-day increase in Covid-19 infections. The spike brought the total number of cases in India to 17.3 million, second in the world only to the U.S. More than 195,000 people have died there as of Sunday, though public-health experts believe the death count is likely higher. The U.S. announced Sunday it would send raw materials for vaccines to India and step up funding for the country to manufacture more doses. Ventilators, therapeutics, rapid testing kits and personal protective equipment will be sent as well, the White House said. While vaccine doses were not included, the U.S. is sending a supply of remdesivir, an anti-viral drug used to treat hospitalized Covid-19 patients, a senior administration official said. India’s SurgeIn their Monday phone call, according to senior administration officials, Biden and Modi pledged to work closely together to combat the virus and Biden promised steadfast support for the people of India amid the surge in Covid cases. The aid package includes oxygen and related supplies, with the U.S. looking at sending oxygen generation systems. A team of experts from the Centers for Disease Control and Prevention is being sent to India to assist public-health officials there as well, the White House said.
Anthony Fauci, Biden’s chief medical adviser, said Monday that “everything is on the table right now for discussion” when he was asked why the U.S. had not given away doses of the AstraZeneca vaccine, including to India.
“I think we can safely say that the issue of doses of vaccine, particularly the AZ vaccine, are certainly under active discussion. And I would say, safely, that everything is on the table right now for discussion,” Fauci said at an event hosted by the Harvard School of Public Health. The U.S. has used wartime powers and contractual clauses to give its orders priority — meaning that there are, as of now, no known U.S.-made doses that have gone to any buyer other than the U.S. government. A spokesman for AstraZeneca said the company can’t comment on specifics but said the doses were part of AstraZeneca’s supply commitments to the U.S. government, and decisions to send supply to other countries would be made by U.S. officials. Earlier this month, Bloomberg reported that the U.S. stockpile of the AstraZeneca vaccine had grown to more than 20 million doses, part of a total of between 80 million and 90 million doses in some stage of production for the U.S. order. Biden signaled last week that the U.S. was considering loaning more, but said that won’t happen until there is enough supply. “We don’t have enough to be confident to give it — send it abroad now,” he said. “But I expect we’re going to be able to do that.” –With assistance from Jennifer Jacobs, Jeannie Baumann, Riley Griffin and Suzi Ring. from https://ift.tt/3gDXEpp Check out https://takiaisfobia.blogspot.com/
0 Comments
India is in the midst of a humanitarian crisis. The country recorded 352,991 new cases of COVID-19 on Monday, breaking the record once again for the most cases recorded in a single day anywhere in the world since the beginning of the pandemic. Hospital ICUs are overcrowded. There are shortages of oxygen for some patients with the most severe illness, of personal protective equipment (PPE) for those treating them, and of tests to detect the true extent of the epidemic. Read More: ‘This Is Hell.’ Prime Minister Modi’s Failure to Lead Is Deepening India’s COVID-19 Crisis Journalists stationed at crematoria in cities and towns say dead bodies are being burned in far higher numbers than the official statistics of around 1,900 deaths per day over the past week suggest. On Saturday, 20 patients died at a hospital in Delhi after a delayed oxygen delivery. Days earlier at a hospital in western India, 22 critically ill patients died when an oxygen tank ruptured. 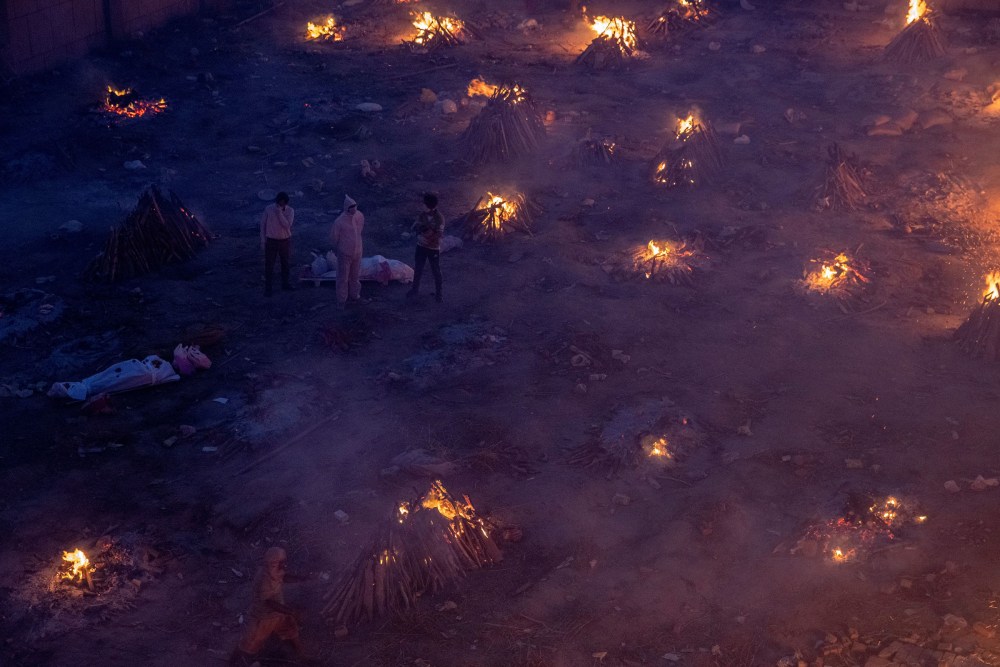
Where India needs the most helpIndia’s health ministry says that the country has the oxygen it needs, but is facing bottlenecks in the transportation from facilities in industrial locations to hospitals where it is needed. On Sunday the Indian government approved funds to set up 551 new oxygen generators at hospitals across the country. Special trains carrying oxygen, which is difficult to transport due to its high flammability, have also been sent to shortage-hit cities. But there are other shortages, too. India is facing scarcity of critical drugs for treating serious cases of COVID-19 including Remdesivir and sedatives for patients on ventilators. Authorities have warned that the shortages have been made worse by concerned citizens panic-buying. “Hoarding of injections like remdesivir and oxygen in homes is creating a panic and this hoarding is causing a shortage of these medicines,” said the All India Institute of Medical Sciences (AIIMS) on Sunday. The cost of a shot of Remdesivir on the black market has risen from some $12 to $600, the Economist reported. Extra supply would help ease such pressures. There is also a shortage of hospital beds and, more importantly, ventilators needed to treat the most severely ill COVID patients. “India’s health system is on the verge of collapse,” said Ashish Jha, dean of the Brown University School of Public Health, in a Washington Post article Saturday, in which he called on the U.S. to help India set up field hospitals to increase the number of beds available. “At this point medical supplies, oxygen and vaccines are the most important things we need,” says Yamini Aiyar, President of the Centre for Policy Research, a New Delhi think tank. “Multilateral aid will be crucial.” 
How are governments around the world helping?Around the world, governments are waking up to the scale of the crisis. On Sunday the White House said it would begin sending more supplies to India, including ventilators, test kits and PPE. “Just as India sent assistance to the United States as our hospitals were strained early in the pandemic, we are determined to help India in its time of need,” President Joe Biden said in a tweet. On Monday, an Air India plane carrying 328 oxygen concentrators, which pull oxygen from the air and pressurize it into higher concentrations for medical use, landed at New Delhi’s Indira Gandhi airport from New York. The U.K., E.U., and even India’s longtime geopolitical rival Pakistan are among others that have also committed to sending supplies including ventilators and oxygen concentrators. Read More: How the Pandemic Is Reshaping India The White House also said it would overturn a ban on the export of raw materials used in vaccine production. “The United States has identified sources of specific raw material urgently required for Indian manufacture of the Covishield vaccine that will immediately be made available for India,” a U.S. National Security Council spokesperson said in a statement. In addition, the U.S. is also sending funds to BioE, India’s main vaccine manufacturer, with the aim of helping it produce at least 1 billion vaccines by the end of 2022, the statement said. India is lagging much of the rich world in overall vaccination rate. More than 109 million Indians have received at least one dose already, but for a country with almost 1.4 billion people that’s less than 10% of the population. More than 41% of the total population in the United States have received their first dose. 
How individuals are trying to helpAlthough a crisis of this scale requires state intervention, people in India and around the world are using the internet to provide much-needed help to stricken families and on-the-ground aid groups. As the virus swept through the country in recent weeks, many Indians took to Twitter to share the stories of people in need of oxygen, or ventilators, or a hospital bed, in the hopes of finding help online. Many, like the journalist Vinay Srivastava who live-tweeted his final moments in need of oxygen, were unsuccessful. Others have been sharing reputable organizations engaged in work on the ground in the hopes of encouraging those who can to support those efforts. Some of these organizations, like Give India, have hubs where you can choose to donate money to vetted funds for the purchase of life-saving medical equipment, food for families dealing with hunger, or reusable sanitary pads for women and girls who have gone without because of pandemic pressures. Each accepts foreign donations. Activists have also compiled a Google Doc containing a long list of crowdfunders, including specific families in need, funds specifically targeted at alleviating the burden on minority communities, and mental health support for those impacted. (TIME has not vetted the donation details in this document.) In a state of crisis, India needs as much help as it can get. And when continued spread means a higher probability of viral mutations arising too, helping India is not just a vital humanitarian decision: it’s one of the best ways to help the world beat COVID-19 for good. from https://ift.tt/3u0TsUQ Check out https://takiaisfobia.blogspot.com/ As the U.S. Moves Toward Post-Pandemic Life COVID-19 Is Still Devastating the WorldEspecially India4/26/2021 The pandemic won’t end for anyone until it ends for everyone. That sentiment has been repeated so many times, by so many people, it’s easy to forget it’s not just a cliche—particularly if you live in one of the wealthy countries, like the U.S. and Israel, that has made significant moves toward what feels like an end to the COVID-19 era. Israel, for example, has fully vaccinated more than half of its population and about 90% of its adults 50 and older are now immune to the virus—enough that the country is “busting loose” and “partying like it’s 2019,” as the Washington Post put it last week. The U.S. is a bit further behind, with nearly 30% of its population fully vaccinated, but the possibility of a post-pandemic reality is already coming into focus. While daily case counts remain high, they are far lower than they were even a few months ago—about 32,000 diagnoses were reported on April 25, compared to daily tallies well above 250,000 in January. Deaths have also trended downward for most of 2021. The U.S. Centers for Disease Control and Prevention has relaxed its guidance on travel and indoor gatherings, and some states have repealed mask mandates and other disease precautions. But while people in certain affluent countries celebrate a return to vacations and parties, COVID-19 remains a dire threat in many nations around the world—nowhere more so than India. For five days in a row, the country has set and reset the global record for new cases in a single day, tallying about 353,000 on April 26. By official counts, about 2,000 people in India are dying from COVID-19 every day as hospitals grow overtaxed and oxygen supplies run short. Experts say the true toll is likely even higher than that. People are dying as they desperately seek treatment, and crematoriums nationwide are overwhelmed. It can be difficult to grapple with that devastating reality when people in countries like the U.S. are reuniting with loved ones and cautiously emerging from lockdown. How can both scenarios be happening at once? The answer, as it often has during the pandemic, lies in disparity. As of April 26, 83% of vaccinations worldwide had been given in high- and upper-middle-income countries, according to a New York Times data analysis. In the developing world, many countries are preparing for the reality that it could take until 2022 or even 2023 to reach vaccination levels already achieved by richer countries today. Even in India, one of the world’s leading vaccine manufacturers, fewer than 10% of people have gotten a vaccine—a cruel irony, as people in India die in the streets while those thousands of miles away celebrate receiving their second doses. To truly defeat COVID-19, we must reckon with that cognitive dissonance, says Dr. Rahel Nardos, who is originally from Ethiopia and now works in the University of Minnesota’s Center for Global Health and Social Responsibility. As an immigrant and global health physician who lives in the U.S., Nardos says she inhabits two worlds: one in which the U.S. may feasibly vaccinate at least 70% of its population this year, and another in which many countries struggle to inoculate even 20% of their residents in the same time frame. “It’s a huge disparity,” Nardos says. “We need to get out of our silos and start talking to each other and hearing each other.” That’s imperative, first and foremost because it could save lives. More than 13,000 people around the world died from COVID-19 on April 24. Remaining vigilant about disease prevention and monitoring, and working to distribute vaccines in countries that desperately need them to fight back COVID-19 surges, could help prevent more deaths in the future. That’s especially critical for developing countries, many of which are so overwhelmed by COVID-19 that nearly all other aspects of health care have suffered. “We may be looking at five, 10 years before they can get back to their baseline, which wasn’t that great to begin with,” Nardos says. There’s also a global health argument for distributing vaccines more equitably. Infectious diseases do not respect borders. If even one country remains vulnerable to COVID-19, that could allow the virus to keep spreading and mutating, potentially evolving to such a point that it could infect people who are vaccinated against original strains of the disease. Already, vaccine makers are exploring the possibility of booster shots to add extra protection against the more transmissible variants currently circulating in various parts of the world. We aren’t at that point yet; currently authorized vaccines appear to hold up well against these variants. But if the virus keeps spreading for years in some areas, there’s no telling what will happen, says Jonna Mazet, an epidemiologist and emerging infectious disease expert at the University of California, Davis. “Evolution of those new strains could go into multiple directions. They may evolve to cause more severe or less severe disease. Some of the variants [could be] more concerning for young people,” Mazet says. “The whole dynamics of the disease change.” And if the virus is mutating somewhere, chances are good it will eventually keep spreading in multiple areas, Mazet says. “Unless or until we have a major shift, we are still going to have large parts of every country that have a susceptible population,” she says. “The virus is going to find a way.” The only way to stop a virus from mutating is to stop giving it new hosts, and vaccines help provide that protection. COVAX—a joint initiative of the World Health Organization; Gavi, the Vaccine Alliance; the Coalition for Epidemic Preparedness Innovations; and UNICEF—was meant to ensure that people in low-income countries could get vaccinated at the same time as people in wealthier ones. COVAX is providing free vaccines to middle- and low-income countries, using funds gained through purchase agreements and donations from richer countries. But supply and funding shortages have made it difficult for the initiative to distribute vaccines as quickly as it intended to. Many of the doses it planned to disseminate were supposed to have come from the Serum Institute of India, which delayed exporting doses in March and April as India focused on domestic vaccine rollout to combat its COVID-19 surge at home. In the meanwhile, many poorer countries have been unable to vaccinate anywhere close to as many people as would be required to reach herd immunity. That will almost surely improve as new vaccines are authorized for use by regulators around the world, and as manufacturers scale up production, but those moves may be months away. COVAX is also developing a mechanism through which developed countries could donate vaccine doses they don’t need. Some wealthy countries, including the U.S. and Canada, have contracts to purchase more than enough doses to vaccinate their entire populations, and have signaled their intent to eventually donate unneeded supplies—but timing is everything. That is, these countries will likely only donate once they are sure their own populations have been vaccinated at a level that ensures herd immunity. On April 25, the Biden Administration said the U.S. would provide India with raw supplies for making AstraZeneca’s vaccine, as well as COVID-19 tests and treatments, ventilators, personal protective equipment, and funding. That’s a significant shift, since the export of raw vaccine materials was previously banned, but it still doesn’t provide India with ready-to-go vaccines. That step may be next, though. The U.S. will export as many as 60 million doses of AstraZeneca’s vaccine once the shot clears federal safety reviews, the Associated Press reports. Gian Gandhi, UNICEF’s COVAX coordinator for supply, says he fears many wealthy countries’ vaccine donations may not come until late in 2021, just when global supply is expected to ramp up. That may cause a bottleneck effect: all doses may come in at once, rather than at a slow-but-steady pace that allows countries with smaller health care networks to distribute them. “We need doses now, when we’re not able to access them via other means,” Gandhi says. The global situation is also critical now. Worldwide, more than 5.2 million cases and 83,000 deaths were reported during the week leading up to April 18. Indian hospitals are so overrun, crowds have formed outside their doors and desperate families are trying to source their own oxygen. Hospitals in Brazil are reportedly running out of sedatives. Iran last week broke daily case count records three days in a row. Countries across Europe remain under various forms of lockdown. Vaccines won’t change those realities immediately—but without them, the global community stands little chance of containing COVID-19 worldwide.
from https://ift.tt/3dTLqY5 Check out https://takiaisfobia.blogspot.com/
Well hello! I’m so glad you’re here. If you’re having trouble viewing this in email, see the TIME.com version here. And, as always, you can write to me at [email protected]. Two weeks ago, I wrote about actor and director Justine Bateman’s new book, FACE: One Square Foot of Skin, in which she asks why women still spend so much time in a frustrating quest to ‘fix’ their faces in response to a culture steeped in anti-aging messaging. ( The response was overwhelming. Hundreds of thousands of you read the piece on TIME.com, and many commented on Twitter or wrote to me. Most praised Bateman for dragging these secret fears into the sunlight, writing: “It’s so great to see Justine advocate for women to be themselves.” And: “The system needs fixing, not women.” Others asked: “How in the hell do we get men to stop caring?” Some feared losing their livelihoods if they didn’t try and look younger. Many men wrote to say they thought Bateman’s unaltered face was beautiful as is; others were less than supportive. Ahem. The book provoked discussions across the internet, so I thought it’d be great to talk to Justine about the reaction and her battle cry of #TheresNothingWrongWithYourFace. Excerpts of our conversation are below and you can see a video here. On the reaction to FACE: One Square Foot of Skin. “I was not prepared at all for the magnitude of the response. I’ve had hundreds and hundreds of DMs on Instagram, saying, ‘I’m so glad someone’s talking about this.’ One woman said, ‘I work as a hospice nurse, helping people to die. And yet I’ve got my co-workers telling me I should do something about my face.’… and ‘it’s made me realize that I need to not be as critical about my face because I have two teenage daughters, and I don’t want them to be sort of tainted with that, that those kinds of thoughts for their own face.’” On anti-aging products. “They are only going to sell us things that they think we need. One very easy way to generate a need for a product is to find what fears already exist and then zero in on those fears. I’m interested in eliminating the buttons that have existed in me or may still exist in me that react to those [fears]. ‘If people think my face is old, then, therefore…’ I think for every woman, there’s a fill in the blank for that. For some, it might be they’re afraid they won’t find a mate for someone else; it might be they’re afraid they won’t, their new business won’t succeed. What I’m proposing is to forget the face. It’s got nothing to do with your face. If you fear you’re never gonna find a mate. That’s a fear that’s going to stay with you for the rest of your life, regardless of what you do to your face. So why address that fear to get rid of it. So that then you can be free of that fear.” On letting go of her younger face. “I always wanted to look eventually, like Georgia O’Keefe. So, all these things that start happening, you know, like, with the loose neck and the hooded eyes and stuff, and I’m like, I’m on my way! I’m gonna make it! … Now you look at my face, and you know what you’re getting… I didn’t dislike my face, but I don’t feel like it represented me the way this face represents me, you know what I mean?” On people saying she looks ‘old.’ “When people say something like that, they’re telling me about themselves. They’re not telling me about me. They’re telling me about how they look at life, and they’re telling me how they look at themselves.” On ageism. “If somebody is running a firm, and the woman looks older, and they don’t like that, it’s an interesting thing to dig into. What do we think is going on in that man, or that woman’s head, that they don’t like that they have an employee that looks older? I mean, we can guess. It might be the rule thing, like, well, she’s not doing anything about her face. And that’s the rule you should, and she’s breaking it. It could be: ‘Oh, I’m afraid that [an older face] indicates that we’re not innovative anymore. And I don’t want other companies to imagine we’re not innovative. And so I’ve got to get rid of that evidence,’ which is their fear. I believe then that individual has a fear that their company is not innovative. He imagines, ‘Oh, I can do this easy fix where I get rid of an older person.’ And then, ‘Okay, I’ve solved that problem.’ But he can have everybody at the company be 22, and he will still have a fear that his company’s not innovative.” On feeling that we can’t be relevant if we look older. “I’m just saying, maybe it’s not your face. Maybe it’s the direction you’re looking. Maybe there’s a new direction open for you, and you’re not paying as much attention to it. And the timing’s right for you to go in that direction. Because I feel like everyone’s got a basket of opportunities, and skills and talents that come with you, when you’re born. And I feel like that basket does not care what gender I am, what, what the state of my face, my age, it doesn’t care. You can’t steer a parked car, right? But if you start moving it, you’ll start seeing that there are certain opportunities that you hadn’t expected before, maybe in directions that hadn’t occurred to you. To me, I think life is fun. You have uncomfortable things and comfortable things, but they’re all conspiring to make you more and more yourself, one more who you are.”
Subscribe here to receive an essay from @susannaschrobs every Sunday. Road Trip Update! Thanks to all of you for the good wishes and kind invitations for my cross-country voyage. I’m flipping the script a little bit. Dog and I will now be traveling West to East at the end of May, and I’ll be posting from the road on Instagram @SusannaSchrobs COPING KIT ⛱There’s a Name for the Blah You’re Feeling: It’s Called Languishing Organizational psychologist Adam Gran on the neglected middle child of mental health can dull your motivation and focus — and it may be the dominant emotion of 2021. Did someone forward you this newsletter? SUBSCRIBE to It’s Not Just You here. from https://ift.tt/3gzS47t Check out https://takiaisfobia.blogspot.com/ In a 10 to 4 vote, a committee of the U.S. Centers for Disease Control (CDC) on April 23 decided to lift the temporary hold on using the COVID-19 vaccine made by Johnson & Johnson/Janssen. The U.S. Food and Drug Administration (FDA) and CDC had temporarily halted vaccination with the J&J shot on April 13 after six reports of unusual blood clots in the brain occurred among nearly 7 million people vaccinated with the shot in the U.S. The panel of 15 independent experts was under some pressure to make a decision about the safety of the vaccine, which is the only single-shot vaccine for COVID-19 currently authorized in the U.S. But after an initial meeting on April 14, the group decided it needed more information. Over the past week, an additional nine cases of unusual blood clots, called cerebral venous sinus thrombosis (CVST) were reported, bringing the total so far to 15 cases. All 15 have occurred in women, and most were in their 30s. However, since that’s still among several million people who had recently been vaccinated with the shot, CDC vaccine-safety experts still felt the overall risk is very small. To put those 15 cases in context, Dr. Sara Oliver, from the CDC’s National Center for Immunization and Respiratory Diseases, and her team presented data from models they constructed to estimate how the 11-day pause in using the J&J vaccine affected COVID-19 cases, hospitalizations and death. The models showed that if the vaccine were reinstated for all adults over age 18 years, there would be 26 to 45 additional cases of the rare blood clots side effects over the next six months, but 800 to 3,500 fewer admissions to the ICU and anywhere from 600 to 1,400 fewer deaths. If officials restricted the vaccine only to people over age 50—which might make sense given that younger people are more at risk of the clotting side effect— they would expect two to three additional cases of clotting compared to 300 to 1,000 fewer ICU admissions and 40 to 250 fewer deaths. Oliver also broke down the data at the individual level, and reported that for every one million doses of the vaccine administered to women aged 18 to 49—the group at highest risk of the clotting side effect—13 cases of clotting might be expected. J&J representatives at the meeting reiterated that the vaccine starts protecting people against disease within days of administration, and that data from the company’s clinical trials showed it was 85% efficacious in protecting people from severe COVID-19, including against new variants of the virus. They also stressed that theirs is the only authorized vaccine that requires just a single dose, and that it can be shipped and stored at refrigerator, rather than extremely cold, temperatures, making it more accessible to most of the world. “Johnson & Johnson’s vaccine is central to the effort to end the pandemic,” Dr. Joanne Waldstreicher, the company’s chief medical officer told the committee. “Our vaccine provides access to people who are underserved as well as people who typically face barriers in accessing health care services. Our concern is that with continued restriction of the vaccine we are not just delaying vaccination but potentially leaving unvaccinated a portion of the U.S. If the unvaccinated population is large enough, that could risk us reaching herd immunity.” Waldstreich said the company favored adding a warning label about the risk of clotting, similar to the way the risk of anaphylaxis is now included in the label for the mRNA vaccines from Pfizer-BioNTech and Moderna. Throughout the day, members of the committee noted that the data seemed to show the risk of clotting was rare, and that benefits of vaccination with the J&J shot outweighed that risk. But they debated over how to warn people—especially younger women—of the potentially serious side effect. Some favored wording that highlighted the risk specifically, noting that women under age 50 should be given the choice of another vaccine. That’s how the U.K .has decided to roll out the AstraZeneca vaccine, giving people under 30 the choice to get an alternative shot. Other committee members argued that specifically featuring the blood clotting would be inconsistent with the way other side effects for any vaccine, including the other COVID-19 vaccines, are handled; they said that blood clotting risks should be part of the already FDA-approved language, as follows: “Reports of adverse events following use of the Janssen COVID-19 vaccine under emergency use authorization suggest an increased risk of thrombosis involving cerebral venous sinuses and other sites … combined with thrombocytopenia and with the onset of symptoms approximately one to two weeks after vaccination. Most cases of thrombosis with thrombocytopenia reported following the Janssen COVID-19 vaccine have occurred in females ages 18 through 49 years; some have been fatal. Specific risk factors for thrombosis with thrombocytopenia following the Janssen COVID-19 vaccine and the level of potential excess risk due to vaccination are under investigation. Based on currently available evidence, a causal relationship between thrombosis with thrombocytopenia and the Janssen COVID-19 vaccine is plausible…Recipients of the Janssen vaccine should be instructed to seek immediate medical attention if they develop shortness of breath, chest pain, leg swelling, persistent abdominal pain, neurological symptoms (including severe or persistent headaches or blurred vision), or petechiae beyond the site of vaccination.” A lay version of that warning is included in the fact sheets that people who get vaccinated receive when they get their shot. Dr. Sarah Long, professor of pediatrics at Drexel University College of Medicine, was one of the four members who voted against the recommendation. “I did not object to the recommendation,” she told the committee in explaining her vote. “I objected to the absence of any kind of guidance from us. I’m very sorry we haven’t chosen to put up front the knowledge we have that this unique, clustered side effect is almost certainly related to the vaccine and that there are options.” If CDC director Dr. Rochelle Walensky accepts the group’s recommendation to reinstate the J&J vaccine with an amended warning, then it will be up to the CDC, the state public health departments and individual providers to inform people of the risks of the shot. The CDC is planning on publishing the updated EUA warning both on its website as well as in its weekly MMWR publication on April 27, and fact sheets for providers and people receiving the vaccine will also be updated in coming days. The committee members discussed the burden that would place on some providers, especially those with smaller vaccination programs, who would now, ideally, have to stock two different vaccines in case some people decide against getting the J&J shot. They also considered the possibility that some women uncomfortable with getting J&J shot would end up not getting vaccinated or delaying their vaccination entirely if another option isn’t available immediately. “It places upon us who are making this vaccine available and distributing it, the responsibility to make sure the public is informed about the risk,” said Dr. Jose Romero, professor of pediatrics at University of Arkansas, director of Arkansas department of health, and chair of the committee. “Our responsibility as clinicians is to make sure women understand this risk and when possible, that they have alternatives at the same time that we are administering the vaccine.” from https://ift.tt/2QkLZ4o Check out https://takiaisfobia.blogspot.com/ People Are Reporting Unexpected Side Effects After COVID-19 VaccinationBut Thats Actually Normal4/22/2021 Abigail, a 29-year-old from New York City who asked to use a pseudonym to preserve her privacy, knew to expect some side effects after she got her second Pfizer-BioNTech COVID-19 vaccine in February. The fever, body aches and fatigue she felt were nothing surprising. But when she began experiencing heavy, “stupidly painful, debilitating” menstrual periods, she felt blindsided. “I had not heard a single person talking about it,” she says. While scientists have not confirmed that COVID-19 vaccines can cause menstrual changes like Abigail’s, she feels confident the shot was her trigger, since she experienced something similar after a probable case of COVID-19 last spring. As of mid-April, more than 19,000 people had reported similar experiences in a University of Illinois survey. Even without scientific confirmation, their stories are drawing attention to the fact that vaccination side effects can be far more varied than what’s often discussed in the mainstream. Side effects—while temporarily uncomfortable—are a standard part of vaccination, says Dr. Stanley Perlman, a professor at the University of Iowa’s Carver College of Medicine and a member of the U.S. Food and Drug Administration’s vaccine advisory committee. They show that the body is responding to the shot and mounting an immune response. (Perlman says he considers side effects different from serious adverse events, like the rare blood clots reported in connection with Janssen/Johnson & Johnson’s shot. Most side effects are unpleasant but normal, whereas serious adverse events are potentially fatal issues that demand medical care.) The U.S. Centers for Disease Control and Prevention (CDC) lists common COVID-19 vaccine side effects as pain, swelling or redness at the injection site; fatigue; headaches; muscle pain; chills; fever; and nausea. But that list isn’t exhaustive. In fact sheets describing each vaccine, manufacturers listed additional possible side effects—including diarrhea, joint pain, swollen lymph nodes, hives, rashes and facial swelling—and noted that there could be even more side effects beyond those specified. Menstrual changes may fall into that category, if conclusive evidence links them to the vaccines. (In statements to TIME, a spokesperson for Pfizer said clinical trial participants did not report any menstrual changes. A spokesperson for Janssen said menstrual changes were not tracked as part of its study. A Moderna rep did not return requests for comment by press time.) There have also been reports of people developing rashes and other skin conditions, like so-called “COVID arm,” after vaccination. Others have experienced brain fog that feels akin to a marijuana high. In rare cases, people have also developed shingles after COVID-19 vaccination, as small numbers of people do after taking other types of drugs, but those findings are preliminary. There are bound to be more side effects observed and reported by the general population than are uncovered during a clinical trial, Perlman says. Each COVID-19 vaccine trial had to have at least 30,000 participants. That’s a lot, but it’s a small fraction of the 215 million people who have now been vaccinated with at least one dose in the U.S. alone. Even side effects that were uncommon during clinical trials may affect a relatively large number of people once vaccines are widely distributed, Perlman says. “If you have a trial where you’ve looked at 30,000 people and something happens to 0.1% of them, that’s 30 people,” Perlman says. “But then when you change that 30,000 to 30 million, now there’s many more people having the same side effect.” Not everything that happens post-vaccination is caused by the vaccine, of course. When millions of people get vaccinated every single day, some are bound to experience health problems following but unrelated to the vaccine, Perlman says. Even still, it’s important for people to monitor their health in the days following vaccination and alert their doctors to anything out of the ordinary, so health authorities know which side effects to emphasize or look into further, says Paul Duprex, director of the University of Pittsburgh’s Center for Vaccine Research. Members of the public can report their side effects using the CDC’s Vaccine Adverse Event Reporting System or V-safe tracker. “Those voices matter,” Duprex says. “Patterns can only be recognized [if people who get vaccinated] voice what happened and people who hear those voices collect and collate” the data. There’s a tendency among researchers to discount observations if their cause can’t be proven, Duprex admits. “There’s a fundamental difference between correlations and a cause,” he says. But “none of those things should be dismissed.” Only when researchers have all the data can they adequately assess what’s really happening and communicate that to the public. “There are things that we can only learn,” Duprex says, “if those vaccinated voices are heard.” That’s what people like Abigail are trying to make sure of, by speaking out about potential side effects like menstrual changes. Abigail says she remains glad she got vaccinated, and recommends that everyone do the same. She just wants others to go into the experience feeling more informed than she did. from https://ift.tt/3xgYTB2 Check out https://takiaisfobia.blogspot.com/ Survey: Faith-Based Approaches Could Help Convince Millions of Americans to Get COVID-19 Vaccines4/22/2021 A new survey released by the Public Religion Research Institute (PRRI) and the Interfaith Youth Core found that COVID-19 vaccine hesitancy remains prevalent in some American communities—but a faith-based approach could prove crucial in the fight to combat it. The survey sampled over 5,600 adults across the U.S. between March 8 and 30. It found that among Americans who attend some type of religious service at least a few times a year, 44% of people “hesitant” about COVID-19 vaccines said that a faith-based approach—or approaches—could impact their eventual decision to get vaccinated, and 14% of people “resistant” to the vaccine said the same. (Across all people surveyed, regardless of religious affiliation, 26% of “hesitant” Americans and and 8% of “resistant” Americans said that faith-based vaccination campaigns and public health drives would make them more likely to get the shot. The results suggest faith-based approaches could potentially help sway millions of Americans into getting vaccinated. The approaches that the surveyed people were asked to consider included a respondent’s religious leader or fellow religious community member receiving the vaccine, a religious leader encouraging the community to get vaccinated, a religious community holding an informational forum on the vaccine, a nearby congregation serving as vaccination site or their religious community providing help to arrange vaccine appointments. 
As of Monday, all adults in America are eligible to get the COVID-19 vaccine. At least 134,400,000 people have received at least one dose, and at least 87,500,000 are fully vaccinated, according to the U.S. Centers for Disease Control and Prevention. Still, the pace of vaccination is varying demographically. The U.S. has reached a transition point, argues Robert P. Jones, CEO and founder of PRRI, where communities need to implement flexible and personalized strategies to convince people to get the potentially life-saving shots. “We’re going to need every tool in the toolbox, I think, to get us to herd immunity,” he continues. “And what we’re showing is that [faith-based approaches] can really be a key part of the solution, I think particularly for some of these communities that have been very hesitant but have close ties to religion.” Some faith leaders have already launched pro-vaccination campaigns within their communities, and PRRI’s results suggest those campaigns might well be working, says Jones. While PRRI found that 32% of Black Protestants said they were hesitant to get the vaccine, and 19% said they wouldn’t get it at all, the survey also found that attending religious service was positively correlated with vaccine acceptance in the community. Fifty seven percent of Black Protestants who attended church services at least a few times per year said they’d take the vaccine, while only 41% of Black Protestants who don’t attend services said the same. Read more: How One South Carolina Pastor Is Combating Vaccine Hesitancy in Communities of Color PRRI’s survey found the opposite was occurring among white evangelical Protestants, though to a smaller degree: 43% of white evangelical Protestants who regularly attended services said they’d take the vaccine, compared with 48% of those who attended church services less frequently. But the survey also found that a pro-vaccination campaign could have a huge impact in the community. Among white evangelical Protestants who attend services at least a few times a year and are hesitant to take the vaccine, 47% said a faith-based approach would make them more likely to get vaccinated. “So there’s actually a great amount of untapped potential among white evangelical churches here for… faith based interventions,” says Jones, “even though we’re not seeing it quite happening right now.” Pro-vaccination campaigns could also prove impactful in other communities, the survey found. Thirty three percent of Hispanic Americans who said they were hesitant to take the vaccine said a faith-based approach would make them more likely to get the shot, as did 26% of Republicans and 24% of rural Americans. “To overcome hesitancy you really do need [all kinds] of cultural resource,” Jones adds. “And religion is, I think, an often overlooked resource in this space, but one that we show can play a pretty critical role.” from https://ift.tt/2PgZbqm Check out https://takiaisfobia.blogspot.com/ Over the past year, rising deaths from COVID-19, police brutality, anti-Asian hate crimes, and the inequitable damage of climate breakdown, have made the manifold harms of racism easier for everyone to see. Harms that were once shielded from public consumption by segregation or shrouded from public scrutiny by stories depicting the U.S. as a nation of fairness and freedoms, are now the center of an ongoing national confrontation with racism and its impacts on health, safety, and justice. Yet amid growing calls for anti-racism and health equity, troubling reports have emerged highlighting the ways the U.S. health care industry avoids even talking about, let alone addressing, racism. Few know just how extensive this problem is. Our new report published in Health Affairs examined the top four medical journals in the world and found that they almost never publish scientific articles that name racism as a driver of poor health outcomes. Of the more than 200,000 total articles published over the past 30 years in the New England Journal of Medicine (NEJM), The Lancet, the Journal of the American Medical Association (JAMA), and British Medical Journal (BMJ), less than 1% included the word “racism” anywhere in the text. And among the few articles that did, upwards of 90% were predominantly opinion pieces, not scientific investigations. Just think about that. In a field that maligns anecdotal evidence as the least rigorous and most untrustworthy, our examination found that the primary “evidence” accepted and published about racism were people’s scholarly opinions. Brilliant as those opinions may be, medicine is purportedly built upon the rigor of empirical scientific investigations, not the substance captured in commentaries. But when we looked at the science published on racism in the four leading medical journals in the world, we found it was thin, to say the least. Among these top medical journals, during the past 30 years, each managed to publish as few as four and at most 29 scientific studies that included the term “racism.” Moreover, most of these empirical investigations were solely descriptive in nature. In other words, the studies would describe a gap in health outcomes between racial groups but never empirically test if or how racism contributed to the gap and never determined the effectiveness of potential interventions. In fact, you could count on one hand the articles that attempted to even measure forms of racism as potential drivers of a health outcome. For the New England Journal of Medicine, arguably the most influential medical journal in the world, you only need a single finger. Perhaps most notably, JAMA—a journal recently thrust into public discourse after its editor-in-chief was placed on administrative leave for sponsoring a podcast that denied the existence of structural racism in medicine—has not published a single empirical study measuring racism in the 30 years we looked at. Yet, racism is neither a niche topic nor a rare exposure: 40% of the U.S. population are people of color (i.e., are not “white non-Hispanic”), research illustrates that experiences with racism are common, and the impacts of structural racism on health are well-established. Indeed, there is nearly 200 years of knowledge connecting racism to poor health outcomes. And other credible scientific journals are publishing plenty of empirical studies directly investigating the impact of structural, institutional, interpersonal, and internalized racism on health. But you would never know that by reading today’s preeminent medical journals. That alarms us. Because in medicine, nearly everything we do is based on evidence. And the evidence that is weighed most heavily are the data published in our field’s top journals. Screening protocols, medication recommendations, and basic knowledge about diseases and treatments hinge on the “evidence.” This “evidence,” or compilation of studies published in the medical literature, shapes how society comes to understand the causes of health problems and effective treatments. As a result, the “evidence” is taught in schools, from elementary classes to graduate level clinical training. It is used to inform public policy, set public health guidelines, adjust government budgets, prioritize the nation’s research agenda, determine standards of medical care, and decide which treatments insurance covers. Therefore, when studies can illustrate the links between, for example, residential segregation, exposure to air pollution, and chronic illnesses like asthma, they also help elucidate potential , like changes to housing policies, transportation policies, and increasing green space. Or when empirical investigations can expose the connections between police killing unarmed Black and Latinx civilians and both mental health and educational outcomes in Black and Latinx youth they also highlight how policing reforms can address mental health and educational achievement gaps. Conversely, when the world’s leading medical journals fail to publish scientific evidence about how racism harms health, it can have a devastating impact on how health systems, governments and public health infrastructures serve people of color and fund research on racial health inequities. In addition, if we, as medical and public health professionals cannot name and confront racism as a root cause of racial health inequities, it profoundly affects what the broader public knows and doesn’t know about the racial distribution of health and disease and its social causes. When it comes to racism, ignorance is not benign. For example, throughout these journals’ history, some of which date their origins as far back as the 1800s, nearly all of the editors and chiefs have been white men, with a few exceptions. For this group, who act as gatekeepers of scientific knowledge, failure to publish about the effects of racism can contribute to power asymmetries within health careand limit what is known about racism and health. In regards to the latter, this has contributed to attempts to refute the role of racism and white racial dominance in shaping health and health care and to argue that racial inequalities in COVID-19 are driven by genetic differences between racial groups . Recent essays published in these leading medical journals have also falsely claimed racism is just too messy to measure and that racial groups reflect genetically distinct populations. As the grossly unequal racial distribution of COVID-19 case rates, death rates, and vaccination rates continue to reveal, racism structures who has access to vital health care services, adequate workplace protections, sufficient housing, safe parks, and clean air—all of which together structures who lives and who dies early in this pandemic. Consequently, in the past year alone, while COVID-19 caused U.S. life expectancy to fall by one year, the life expectancy of Black men and women fell by three and 2.3 years, respectively. In short, racism threatens and shortens lives. It is a public health crisis. And at this point, attempts to ignore that reality are a choice. On March 1, the U.S. National Institutes of Health announced new initiatives to address the impacts of “structural racism on biomedical research.” On April 8, the U.S. Centers for Disease Control and Prevention declared racism “a serious public health threat.” It is past time for the world’s leading medical journals to name racism, publish evidence on how racism harms health, and articulate how dismantling racism can prevent racial health inequities. from https://ift.tt/3dAf3NY Check out https://takiaisfobia.blogspot.com/ Katherine McConnell wanted to make sure that she and her employees didn’t fall back into their old habits when they returned to the office in Sydney, Australia—where the coronavirus situation has stabilized—after several months of working from home. So McConnell, the founder and CEO of financial technology company Brighte, implemented a flexible working policy, allowing employees to continue to work from home even after the office reopened. Still, she found herself rushing between meetings and spending increasingly long hours in the office—and missing things like eating lunch with her family. So she blocked out one day a week in her calendar to work from home, hoping that would also encourage her employees to follow suit. “As a leader if I don’t show that I can work from home and I will do this, I think that people may copy me and easily return to how they used to do things,” she says, “and I don’t want that to happen, and I know it doesn’t have to.” Many U.S. companies have pushed office return dates to September and beyond. But workers in Australia—where there have been fewer than 30,000 cases of COVID-19 and under 1,000 deaths—are already returning to their offices. That includes employees at Brighte, which specializes in helping homeowners fund home improvement projects, including sustainable energy solutions like solar panels and battery storage. In a video interview with TIME, McConnell shared how Brighte is managing flexible work—and what lessons it can offer to companies elsewhere as they navigate their own return to office life. That might be helpful in a world where more than 70% of people want to split their post-pandemic time between in-person and remote working, according to a PwC survey of 32,500 participants from 19 countries released in March. Brighte, which has about 115 employees in Australia’s largest city of Sydney (as well as a smaller office in the Philippines), went into work-from-home mode in March 2020 as COVID-19 cases spiked. But the country of 26 million people kept the virus in check with strict lockdowns and stringent border controls. As of April 20, there are just three active locally-transmitted COVID-19 cases in the state of New South Wales, where Sydney is located. Brighte employees were able to start returning to the office by June of last year, although the office looked and felt different—desks were spaced farther apart, meeting rooms had lower maximum capacities, and hand sanitizer dispensers were installed throughout. (Some of these precautions have since been eased given the loosening of government restrictions and the low number of COVID-19 cases). Some employees of the company—which in 2019 was ranked the fourth-fastest growing company in the Asia-Pacific region in the Deloitte Technology Fast 500—were nervous to go back to the office. So company leaders let them know that there’s flexibility to meet their needs. “We have an accommodation and an acceptance that people will work from home one to two days a week,” says McConnell. But some people work remotely more than others, and she’s empowered her management team to make decisions that work for their direct reports. “It’s an individual basis, it’s a team-by-team basis, it’s high levels of communication, and it’s a delegation of authority to team leaders and team managers to work with their team to understand their individual requirements.” Some things have shifted to make that work. McConnell tries not to schedule company-wide meetings and social events on days like Friday, when more people tend to work from home. Meeting invites include an option to join by Zoom, and if someone specifically wants to host an in-person meeting, they let everyone know in advance. On busier days, between 70-80% of employees work from the office. She says that on quiet days, like Mondays and Fridays, about 10 to 15% of staff show up in person. That, McConnell says, draws people to the office who want quiet time to think or do difficult work. “People think about the office space differently knowing that it’s normally an open-plan, busy office that’s noisy, and so you’re actually able to create different spaces for your people to work,” she says. But it also means that she has to remember not to default to pre-pandemic thinking. “It is interesting seeing it empty,” she says. “Sometimes you can default to, ‘where is everyone?'” “That’s the old thinking, and I think that’s the thinking that we know is not true,” she adds. “Just because I can’t see everyone on my team doesn’t mean that they’re not working, doesn’t mean we’re not delivering what we need to.” Remote work has presented other challenges, too, which will likely sound familiar to those logging in from home across the world. McConnell says her company is trying to keep an extra eye on employees’ mental health, for instance. “It’s hard to switch people off,” she says. “I really think that it changes the onus on the employer now to really think about their employees health, like their mental health, their long-term ability to perform, not just short-term.” But overall, she says that her employees are adjusting well to the new normal. “They’re loving the ability to be able to manage their life and manage their work,” she says. “They just seem happier because they have balance in their life.” from https://ift.tt/3niFISR Check out https://takiaisfobia.blogspot.com/ Will Trust in the Johnson & Johnson Vaccine Recover? Europes AstraZeneca Experience Suggests Not4/20/2021 When the U.S. Food and Drug Administration and Centers for Disease Control and Prevention recommended stopping use of the Johnson & Johnson/Janssen COVID-19 vaccine on April 13, they declared the action a “pause”—a brief intermission as the government investigates a possible link between the vaccine and blood clots in a small number of recipients. The agencies may lift that recommendation as soon as this week, and vaccination with the Pfizer-BioNTech and Moderna shots has continued. However temporary it might be, a recent YouGov/Economist survey suggests that the J&J pause has already hurt U.S. public trust in the vaccine: Before the announcement, 52% of respondents said that the J&J shot was safe, compared with just 37% after the pause. (Trust in the Pfizer and Moderna vaccines appears unaffected, and a separate Axios-Ipsos poll found that most Americans believe the pause was the right move.) Those findings have fueled a debate among scientists, researchers and others: is it wise to pause a vaccine’s use after only six blood clotting cases were identified after 6.8 million shots were distributed, given the potential blow to public trust? To help answer that question, we can look to Europe. After France and Germany temporarily halted use of the AstraZeneca-Oxford University vaccine over similar blood clotting issues in March, skepticism of that shot among residents has increased precipitously, according to YouGov polling. Meanwhile, in the United Kingdom—where use of the AstraZeneca vaccine was limited by age, rather than paused entirely—distrust has held relatively steady. If the J&J pause reveals key new data about the vaccine’s safety, it may prove worthwhile. But Europe’s experience offers warnings about the potential damage such a halt can cause. Obviously, the U.K., Germany, France and the U.S all have different on-the-ground realities, but each offers a valuable case study into the potential consequences of a vaccine pause in terms of public trust. United KingdomIn the U.K., confidence in the AstraZeneca vaccine took a minor hit after the blood clotting reports first emerged, but the British are still significantly more likely than the French or the Germans to see that shot as safe. Why? For one thing, the country “went all stops out” to vaccinate as many people as possible from the start, says Heidi Larson, the founding director of the Vaccine Confidence Project at the London School of Hygiene & Tropical Medicine. For example, public health officials decided to delay recipients’ second doses in order to administer first doses to more people—an untested approach that, for now, appears to have paid off. The speed of the U.K. rollout helped it build momentum, experts say. Moreover, as more people get inoculated and very few have severe side effects, those who felt cautious at first may eventually be convinced to get vaccinated themselves, says Scott Ratzan, a professor at the CUNY Graduate School of Public Health and Health Policy and the editor-in-chief of the Journal of Health Communication. AstraZeneca’s vaccine has also had slightly more time to succeed in the U.K. compared to other European countries. The British authorized the shot on Dec. 30, a month before the E.U. Accordingly, the shot has made up a relatively large proportion of all shots given in the U.K. (As of April 5, more than 20.6 million people there have received their first AstraZeneca shot, compared to 11 million Pfizer-BioNTech recipients). Furthermore, AstraZeneca’s shot was developed in the U.K., giving it a boost in the British public eye—for Brits, the vaccine “has been the pride of their country,” says Larson. But perhaps most significantly, the U.K. had a unique response to the AstraZeneca concerns: rather than pause use of the vaccine entirely, British regulators on Apr. 7 restricted its use to people over the age of 30, as the clotting issues appeared to be a more serious concern for younger recipients. Polling data suggests that decision led to a relatively minor increase in distrust of the shot—in an April 7-8 YouGov poll, 13% of people in the U.K. said they viewed the AstraZeneca vaccine as unsafe—only a slight uptick from the 9% who said so in a previous poll conducted March 15-16, before the age restriction was issued. GermanyBack in June of 2020, Germans were primed and ready to be vaccinated—about 68% said they would get a vaccine that’s “proven safe and effective,” compared to about 72% of Brits, according to a poll conducted at the time by Larson, Ratzan and other scholars published in Nature. However, Germans’ confidence in the AstraZeneca vaccine began flagging around the time Germany announced its pause on March 15: in a YouGov poll conducted March 15 to 16, just 32% of Germans said the AstraZeneca vaccine was safe, down from 42% a month before. As of April 18, Germany has administered about 17.6 million doses of the Pfizer vaccine, nearly 6 million doses of AstraZeneca, and about 1.8 million Moderna doses. The AstraZeneca vaccine got off to a relatively slow start in Germany—the European Union didn’t authorize it until February, almost six weeks after approving Pfizer shots. Even after Germany began using AstraZeneca’s vaccine, confusion plagued its rollout. For instance, while European regulators initially recommended its use for anyone over 18, German officials said it should be given exclusively to people under 65, arguing that there was not enough data to support using it in older people. It took until March 4 for Germany to recommend AstraZeneca’s use in those over 65. The key difference between Germany and the U.K. is how Berlin handled the AstraZeneca blood clotting reports. While the U.K. only limited AstraZeneca’s use by age, Germany on March 15 paused the vaccine’s use entirely. German Health Minister Jens Spahn said the decision was a “precaution” meant to give regulators time to investigate the issue. Germany announced it would start using the AstraZeneca vaccine again just three days later—though on March 30, the country tweaked its recommendations yet again, restricting its use to those over 60. Furthermore, while the AstraZeneca shot may have had home court advantage in the U.K., its origin may have been a liability in Germany and elsewhere in Europe. The vaccine rollout was one of the U.K’s first major actions since leaving the European Union in January of last year, a move that sparked resentment around the bloc, and may have set the stage for E.U. distrust of the U.K. when it came to vaccination strategy. European Commission President (and German national) Ursula von der Leyen criticized the U.K.’s decision to authorize the AstraZeneca vaccine before Europe had, saying in early February that the country had compromised “safety and efficacy.” Another, completely separate factor worth considering: on Jan. 25, the German newspaper Handelsblatt published an article claiming that the vaccine was only 8% effective in people over 65—an assertion that was swiftly debunked, but generated international headlines nonetheless. These developments, especially the full pause, may have tarnished the shot’s reputation among Germans. That, in turn, could be a warning sign for the U.S.—Ratzan, for instance, warns that it could be a “real challenge” to restore Americans’ confidence in the J&J vaccine. “If [recipients] have a choice of other vaccines, they will likely want to take a two-dose vaccine that they believe is safer, that never has been paused, than a single-dose vaccine that may have a very, very small risk,” he says. FranceAs it did in Germany, the AstraZeneca vaccine got off to a late start in France—it wasn’t distributed there until Feb. 6, about six weeks after Pfizer-BioNTech and Moderna vaccinations began. Also as in Germany, French regulators have flip-flopped on their age recommendations. And, most crucially, France, like Germany, fully paused AstraZeneca use for several days amid the clotting reports. As of April 18, France had administered about 12.3 million Pfizer doses, 3.4 million AstraZeneca doses and 1.3 million Moderna doses. But unlike Germany, France was “very fragile ground” for a new vaccine to start with, Larson says. In 2016, France was found to be the most vaccine hesitant out of 67 countries surveyed, with 41% of respondents saying that they do not feel vaccines are safe, compared to a global average of 13%, according to polling conducted by Larson and others and published in The Lancet. In a similar Nature poll conducted this past June, only about 59% of the French said they wanted a COVID-19 vaccine even if it was proven safe and effective, compared to 71.5% of respondents overall across 19 countries. Experts say French distrust in vaccines has been fueled by a number of health-related scandals. After an unpopular swine flu vaccine initiative in 2009, for instance, 38.2% of the French population said they distrusted vaccines, compared to 9.6% beforehand. The French government has also been criticized for bungling the vaccine rollout. Bureaucratic obstacles aimed at ensuring that people give consent for the vaccine, including mandatory consultations for the shot, have hindered speedy vaccination. Political messaging has been an issue, too. For instance, on Jan. 9—the same day European regulators approved the AstraZeneca shot—French President Emmanuel Macron incorrectly described it as “quasi-ineffective” for people over the age of 65. While he later said that he would be willing to take the shot, Macron’s comment “certainly didn’t help” vaccine confidence, says Larson. Larson is quick to emphasize that no one factor has been the root cause of vaccine hesitancy in any given country. However, the U.S. should be wary of the way uncertainty over a vaccine’s status or safety can open the door for hesitancy to spread. “I truly and deeply hope that a pause is genuinely a pause, and it doesn’t get into weeks and a month, because it will really undermine public confidence,” says Larson. “If there’s one message to the U.S. out of all this, it’s: don’t let the ambiguity drag on. Because every day just opens the space for misinformation, disinformation, anxiety, and confusion.” from https://ift.tt/3tzsCCZ Check out https://takiaisfobia.blogspot.com/ |
Authorhttps://takiaisfobia.blogspot.com/ Archives
April 2023
Categories |

 RSS Feed
RSS Feed
