|
When Mari and her teenage son, Jesus, were confronted by Border Patrol agents, they did not know they had already crossed the border from Mexico to the U.S. and were now on American soil. To this day, Mari—an asylum seeker from Guatemala—says she doesn’t know where they entered the U.S., only that the agents arrested them and drove them to a facility where dozens of other women were crying. It wasn’t until the agents took Jesus away from her that Mari understood why they were crying. “Do you know that you are committing a crime?” Mari says an agent who worked at the facility asked her. “‘No,’ I told him, ‘I’m running away.'” Mari recalls that the date was May 12, 2018, just five days after then-Attorney General Jeff Sessions announced publicly the implementation of “Zero Tolerance” policy, which resulted in the U.S. government separating more than 5,500 children from their parents at the southern border. The exact number of separated children remains unknown. They took our children. When you don’t have your children, when your children are taken, it feels like dying.The agents told Mari she would be detained at the facility, while her under-age son would be sent to a shelter. They told her they didn’t know when she would see him again. Inside of detention, surrounded by dozens of other women in tears, Mari began to weep. “There wasn’t a single woman who wasn’t crying,” she says in Spanish. “They took our children. When you don’t have your children, when your children are taken, it feels like dying.” Mari, a 48-year-old mother of five, says she fled Guatemala in 2018 to get away from her abusive husband. She took Jesus, her youngest child, and planned to join her daughter, who had arrived in the U.S. more than a decade earlier. Mari has since been reunited with her son and they are both living with her daughter in Santa Ana, California. Her three other grown children remain in Guatemala. Mari spoke to TIME under the condition that TIME only publish her first name, for fear of being located by her husband. Her son’s name Jesus is a pseudonym for the same reason. Mari and Jesus experienced severe trauma from their experience of being separated and detained without knowing when or if they would ever be reunited. Though their family is together again, mental health experts say the trauma of family separation may linger for a lifetime. They are now among over 230 families to request free mental health care as a result of a court order that requires the government to pay for it. Connecting families to mental health servicesIn November 2019, a court ordered the U.S. government to cover the cost of mental health care until January 2021 for any families who experienced separation at the U.S.-Mexico border. The court order came after three immigrant mothers who experienced separation sued the federal government in 2018 on behalf of all separated parents to ask the government to pay for mental health services. Because of the COVID-19 pandemic, that deadline was pushed back to June 2021. Now organizations are working to connect families to these services. In March of this year, Seneca Family of Agencies, a nonprofit organization based in California, was awarded a $14.5 million contract by the government to begin providing free mental health services to the families who experienced separation. Through a program titled Todo Por Mi Familia (All For My Family), Seneca collaborates with several other nonprofit organizations to locate families who could benefit from the court order. Some of those, like the ACLU, Al Otro Lado, Catholic Legal Immigration Network and Kids in Need of Defense, provide legal assistance, while others like Justice in Motion are assisting with the search for parents. “We had all the data, we knew all the information, we knew the ramifications of separating these families, and yet these actions were done,” says Cheryl Aguilar, founder and lead therapist at the Hope Center for Wellness in Washington, D.C., who has provided therapy for families who experienced separation. “We know that once trauma occurs, there’s a mark for a very long time, if not for the rest of your life. That’s what has been done to these kids and these families. The damage is done.”
Paige Chan, the director of Seneca in southern California, says it was clear from the start that her organization would be faced with three primary logistical challenges. The first is finding the parents and children in the first place, she says. The U.S. government often did not keep proper contact information for the parents when they were separated from their children. Roughly 545 parents have still not been located by parties involved with the search. (NBC News reports that the number is higher, with 666 parents remaining at-large). In some cases, parents and children choose not to be reunited in-person, depending on their individual immigration cases. Some parents who have been deported without their children, for example, choose to allow their children remain in the U.S. and pursue a claim for asylum.
The second challenge, Chan says, is building trust with the families, many of whom do not want to be found or contacted by anyone perceived to be associated with the government given their experience of family separation and for fear of being deported. Seneca must also assure families that the services provided are confidential and anything they say to a therapist will not be shared with government officials. Cultural stigmas about mental health care can also create additional barriers to recovery. Third, and last, Chan says, Seneca must build a network of mental health care providers who can provide culturally sensitive therapy in the right language, either in Spanish or in indigenous languages. The therapist could ideally be able to help address the trauma of separation, along with the trauma of the migration journey to the U.S. and whatever pushed the person to flee their country in the first place. The organization is currently working with 230 providers nationwide. “While we can never fully undo the damage done by the separations, we can begin to heal the wounds of these families and bring them back together,” says Ken Berrick, Seneca CEO. Impact of COVID-19Each of those challenges were made worse in March 2020, shortly after the organization finalized their outreach plan, when cases of COVID-19 began to surge in the U.S. “The world was turned upside down and we had to dramatically adjust all the work that we were doing,” Chan says. Work that would previously have been done in person in the states with the highest amount of reunified families to build trust within communities would have to turn remote. Instead of locating families through community programs and going door-to-door to speak to people in person, the Seneca outreach coordinators would have to try and build trust through a phone call. “This is already a population or a demographic that’s going to be really skeptical of strangers calling them for several reasons,” says Johanna Navarro-Perez, program director at Todo Por Mi Familia. “They are perhaps fleeing something in their home country that was challenging or dangerous, and then in this country, at the hands of the government, experienced another sort of intentional trauma… So making that call, being that outreach coordinator, that’s a really tenuous call that you have to be extremely careful and thoughtful about. It’s hard to connect with folks and to be disarming over the phone.” One of those people making calls is Karina Acosta an outreach coordinator at Todo Por Mi Familia. Through contact information provided by lawyers and other nonprofit organizations who are locating the families, Karina makes the phone call and introduces herself. “Nadamas le quería avisar de los servicios que estamos ofreciendo a las familias que fueron detenidas y separadas en la frontera,” she tells them in Spanish, I just want to let you know about the services we’re offering to families who were detained and separated at the border. Depending on how the person responds, Acosta might try to engage them with questions about if their children’s behavior has changed, or share general anecdotes about how other families have benefited from mental health services. Irreparable damageAccording to the American Academy of Pediatrics, which has long spoken out against family separation and child detention, the experience can case irreparable damage to a child’s developing brain, which could lead to both short-term and long-term health impacts. “Even if the family separation did not occur because of the parent—in this case the Administration ripped families apart—a child whose brain hasn’t fully developed and who is not able to make sense of this experience may interpret that information as ‘I was abandoned,'” says Aguilar, who is among the 230 mental health care providers working in partnership with Seneca. Aguilar says that her experience working with immigrants specifically, plus understanding what happens to a child psychologically when they are removed from a caregiver helps inform the treatment she provides. “When a family separation occurs and that attachment is disrupted, what happens when families are reunified is that the families need to be able to have the tools to healthily and happily come back together,” Aguilar says. In many cases, people do want to talk and are ready to and eager to speak to a therapist. Most have accepted Seneca’s offer of free mental health services. But outreach coordinators do find themselves up against cultural stigmas and misunderstandings of mental health care. Cultural stigmasThere are also some families who don’t want to take up the services, or remain skeptical even after speaking with an outreach coordinator. Acosta says she doesn’t pressure families who turn down mental health services—according to court documents 176 families declined services as of November 2020—but she does try to inform families of what mental health services are and that the resource is available until June 2021 if they change their mind. For Mari, accepting mental health services provided by Seneca was easy once she was made aware of what mental health really means, she says. In her household growing up in Guatemala, she says crying was seen as weakness, and could be met with a spanking. But her understanding of therapy changed once she began treatment after being connected with Seneca. More than two years after Mari and Jesus were separated, Mari can recall how she felt panicked, confused and devastated by the separation. Jesus was 15 when they were separated, which lasted a few weeks. During that time they only spoke on the phone twice, both times in tears. When they finally were reunited, they found it a challenge to speak to each other. Now with the help of a therapist, Mari says, Jesus has started to talk to her more and go outside. “I feel like he’s getting better,” she says. “Sometimes, I’d watch him and he’d start crying. I’d ask him why but he wouldn’t tell me. Like he didn’t want to express himself. But the therapist has worked with him, and I see the changes he’s been able to make…I can see that he’s overcoming everything that we have lived.” Mari says she too has benefited from therapy. Her therapist has been able to help her address the separation as well as years of domestic abuse. Mari now uses breathing techniques when she feels sad or overwhelmed, finds it easier to talk about family separation and says she now feels strong after years of internalizing insults from her abuser. “I believed him when he said I was trash. I thought I was trash, and after everything I experienced on the way, everything that happened to me, I believed it and I had bad self esteem,” Mari says. “But I succeeded. I was done, done, done with abuse, I was tired, and I couldn’t take it any more, so I made the journey, and that’s why my therapist tells me I’m very strong.” from https://ift.tt/3m23UXY Check out https://takiaisfobia.blogspot.com/
0 Comments
Moderna is requesting emergency authorization from the Food and Drug Administration (FDA) for its COVID-19 vaccine on Monday — a development the company’s president tells TIME is an “incredible milestone.” The filing for the company’s mRNA-based vaccine is the first step toward getting the shot into hospitals, pharmacies and health centers. The Massachusetts-based biotech is the second company, following Pfizer, to request authorization for a COVID-19 vaccine. Unlike Pfizer’s shot, which is also based on mRNA, Moderna’s vaccine does not need to be stored at ultra cold temperatures in specialized freezers but can remain frozen at -20° C (-4° F), or closer to normal freezer conditions. Once thawed, the doses can be stored in a refrigerator for up to 30 days. That could make Moderna’s vaccine more practical for people in more remote areas where ultra cold storage isn’t available, or where the demand for vaccines isn’t as high, so doses can remain viable for longer periods of time under refrigeration. Read more: Inside the Company That’s Hot-wiring Vaccine Research in the Race to Combat the Coronavirus The submission to the FDA, made up of thousands of pages of data, includes both the final analysis of the phase 3 study of the vaccine, as well as the company’s manufacturing plans. The phase 3 trial, which involved 30,000 people, establishes that Moderna’s vaccine is safe and effective. Of the 196 people in the study who tested positive for COVID-19, only 11 had gotten the Moderna vaccine and the rest received a placebo, meaning the shot was 94.1% effective in protecting people against disease. “That’s almost bang on identical,” to the interim analysis of the data, which was released on Nov. 16, and based on 95 cases, says Dr. Stephen Hoge, President of Moderna. (That data led to a calculation of 94.5% efficacy.) In addition, the company said in the final tally there were 30 cases of severe COVID-19, and all of these cases were among people who received placebo. “We believe the data strongly supports [emergency authorization] for all of the populations tested — adults 18 years and up, of different ethnicities and age groups,” Hoge tells TIME. Read more: Why You May Not Be Able to Get Pfizer’s Frontrunner COVID-19 Vaccine Moderna’s vaccine is based on mRNA, or the genetic code of the COVID-19 virus, which, once injected into the body, acts as the software programming to the body’s immune system hardware, and primes immune cells to recognize and attack the viral genetic material. Prior to the pandemic, mRNA technology was a promising platform for vaccines, but had not led to an approved shot. On Nov. 20, Pfizer became the first company to apply for FDA authorization for an mRNA-based vaccine for COVID-19. The FDA will review that application on Dec. 15. Moderna said that the FDA will likely review Moderna’s application two days later on Dec. 17. Hoge says the company’s vaccine proved safe and effective in a diverse population of people, including African-American and Latinx groups, an important data point since these populations have been disproportionately affected by the pandemic and would likely benefit most from immunization. Moderna developed its vaccine in record time: 63 days after the genetic sequence of the COVID-19 virus was released, its scientists—working with researchers from the National Institute of Allergy and Infectious Diseases—had developed a vaccine ready to be injected into the first study participant. With $955 million in financial support from the Biomedical Advanced Research and Development Authority (BARDA), Moderna is preparing to fulfill its contract with the U.S. government to provide 20 million doses of its vaccine by the end of 2020. Hoge says the company has already produced millions of doses in anticipation of FDA authorization, so that vaccines can start shipping as quickly as possible if that authorization is given. Moderna is manufacturing doses from its Massachusetts facility, but is also preparing another in New Hampshire as well as one in Switzerland to meet its goal of providing 500 million to 1 billion doses globally in 2021. “I personally feel this is an incredible milestone,” Hoge says. The filing represents the pioneering role the company could play in advancing not just mRNA technology but how we develop vaccines against pandemic diseases in the future, he says. “But the goal is to stop the pandemic, and we need to deliver 100 million doses, then the second 100 million doses after that. There is a sense we are still fighting the war,” Hoge adds. “It’s sort of inspiring, and sort of, ‘wow,’ to look back. But at another level if we look forward, we have a lot of work to do, and at least three to six months before we beat this thing, so now is not to time to be too reflective.“ from https://ift.tt/3qcJD4v Check out https://takiaisfobia.blogspot.com/ A version of this article was published in It’s Not Just You, a weekly newsletter by TIME Editor at Large, Susanna Schrobsdorff. Subscribe here to get your dose of small comforts. Well hello! I’m so glad you’re here. This week: The psychology of holiday Zooming, lessons from a recovering pessimist, and a moment of photographic wonder.? Think about Pluto–how it continues to exist as itself, as always, oblivious to human categories. No one else gets to define you or determine your worth. Be a planet despite what they may call you.--Maggie Smith Are You Mad At Me? Show of hands: Who began Thanksgiving by telling a group of beloved family and friends to mute themselves? The great flaw of video platforms like Zoom for non-work gatherings is that only one person (or one little box of people) can talk at a time. This means chaos for people like my people (because no one knows who’s responding to whom). Or authoritarianism (because one of us is liable to appoint themselves the moderator and start calling on people like it’s a city council meeting). But even if you’ve figured out how to have a conversation with 12 people simultaneously, it’s still disconcerting because we lose so much of our nonverbal communication tools in this medium. On Zoom, there are no empathetic side glances, no covertly raised eyebrows, no eye-rolling. And if there is eye-rolling, there’s no way to know who the target is. So naturally, we assume it must be us. This gets us to why group Zooms make us paranoid. Lack of direct eye contact scrambles the emotional radar of humans. You can’t tell if someone’s looking at you, at their dog, or at the door hoping to escape. (To be fair, on Zoom, most of us are probably just staring at our own faces. Scientifically, it’s what we humans love to gaze upon more than anything—even if it’s only to wonder when we got jowls.) Without the body language cues that help us read moods or the kind of connection that happens in just 60 seconds of looking into another person’s eyes we have only our own neurosis to guide us. And that’s never good. Take it from Gianpiero Petriglieri, an Associate Professor of Organisational Behaviour at INSEAD.
And after long periods apart from each other, like during a pandemic, we’re less able to tap a social skill called “theory of mind,” which allows us to intuit others’ intentions and emotions. So if you’ve had a nagging feeling that someone somewhere is mad at you, or less in love with you, or less close to you, you’re not alone. Many of our relationships are happening more in our heads than in real life in this bewildering moment. We construct our own one-sided narratives about who’s thinking what and why. We’re tempted to read way too much between the lines of texts and emails, scanning variances in the volume of emojis, worried that someone’s mad at us. Is she signing off with a single heart now? What happened to the string of balloons and stars? Wait, what month is it? Did I miss something big in his life? But even those maddeningly asynchronous Zoom conversations and texts are a gift–they’re what tethers us to each other across the space-time vortex that is 2020. We get glimpses of each other’s worlds, like a time-lapse sequence of life trundling along despite it all. We can see that the youngest kids are now long-legged people who no longer love purple, someone has a new cat, and a niece who loves to bake made 17 desserts in one week. We find out that we parents haven’t lost our sense of humor yet. That California light is still gold. That no one can make corn pudding like Pam’s. And that the littlest cousin has renamed herself Ruby Starlight. All that goodness ought to hold us for or a bit–maybe even through the rough winter ahead. ? MOMENT OF AWE ✨
Check out the winners of the 2020 Nikon’s Small World Photomicrography Competition. There are images of everything from a chameleon embryo to the Madagascan sunset butterfly’s wing above. And you can get a calendar with this year’s images. COPING KIT ⛱️<strong>Hope was like a garment that I tried on every day. And at first, it was very oversized and itchy and misshapen and uncomfortable, and it didn’t fit at all… But something really strange happened, which is that the more I tried it on for size, the better it fit.</strong> ?Lessons from a Recovering Pessimist Poet Maggie Smith’s new book, Keep Moving: Notes on Loss, Creativity, and Change, is for anyone who’s looking for guideposts on the road to transformation. Check out this interview she did with NPR. <strong>I look at everything in life as borrowed from someone I personally know. Think about it. How do you act if you borrow something from a friend? You don’t get attached, but at the same time, you also take care of it. Live your life like that.</strong> ? How to Let Go of Your Best Laid Plans Writer and stoic Darius Foroux on why we shouldn’t hold too tight to our plans and how to adapt to change. ? Read This Before You Try an Anti-Stress ‘Mood Chew’ Celebrities have been promoting buzzy alternative wellness products, but there’s rarely conclusive evidence that they can help people with mental health challenges. ? Beautiful Distraction: TIME’s Picks for the 20 Best Movies of 2020 It may feel like you’ve watched everything ever made during this long, long year, but TIME film critic Stephanie Zacharek’s picks will surprise and delight. SOME GIVING TUESDAY IDEAS ?One of the best ways to mitigate feelings of helplessness and anxiety is to take some small action–to give help when you feel emotionally needy, to open a door by asking for help. This has never been more true than now as hunger rises in the wake of the COVID-19 pandemic, and more people struggle with mental illness. ☀️Here are a few of my favorite non-profit organizations☀️ The Feeding America networkis the nation’s largest domestic hunger-relief organization. Together with individuals, charities, businesses, and government, we can end hunger. Pandemic of Love is a mutual aid community that connects those who want to give or volunteer directly with those who’ve asked for help with essentials, things like a week’s groceries, gas money, or help with winter coats for kids. The JED Foundation is a nonprofit that protects emotional health and prevents suicide for our nation’s teens and young adults. They partner with high schools and colleges to strengthen their mental health, substance misuse, and suicide prevention programs. COMFORT DOG ??Meet Bucky, the puppy, and her comfort animal. Shared by Maya from New York City. 
?Send questions, requests, and your comforting dog pix to me at [email protected] ? Did someone forward you this newsletter? SUBSCRIBE to It’s Not Just You here from https://ift.tt/33osNFY Check out https://takiaisfobia.blogspot.com/ LONDON — The British government said Friday it has formally asked the country’s medicines regulator to assess whether a coronavirus vaccine developed by AstraZeneca and Oxford University should be authorized for use. The step comes amid questions about preliminary results from trials of the jab, after the company and the university acknowledged that the most encouraging part of their findings stemmed from a dosing error. U.K. Health Secretary Matt Hancock said he had asked the Medicines and Healthcare Products Regulatory Agency to determine whether the vaccine “meets rigorous safety standards.” It’s the second vaccine candidate to reach the formal assessment stage in Britain, following a shot developed by Pfizer and its German partner BioNTech. A third vaccine from U.S. firm Moderna is not far behind. The British government has ordered 100 million doses of the Oxford-AstraZeneca vaccine, and plans to start distributing it in December if it gains approval. The regulator said it could not give a time frame for possible approval of the vaccines MHRA Chief Executive June Raine said “no vaccine would be authorized for supply in the U.K. unless the expected standards of safety, quality and efficacy are met.” Oxford and AstraZeneca reported Monday that their vaccine appeared to be 62% effective in people who received two doses, and 90% effective when volunteers were given a half dose followed by a full dose. They did not mention at the time, but later acknowledged, that a manufacturing issue had resulted in “a half dose of the vaccine being administered as the first dose” to some participants. The drugmakers informed the U.K. regulator of the issue when it was discovered, and it was agreed to complete the late-stage trial with two groups. AstraZeneca has said it plans to conduct a new global clinical trial to assess the vaccine’s efficacy but does not expect that to delay regulatory approval in Britain or the European Union — though the U.S. Food and Drug Administration may take longer. Some scientists have expressed concerns about gaps in the data and the way the results were reported. Only 2,741 people received the half dose, making it hard to know if the effectiveness seen in the group is real or a statistical quirk. A total of 8,895 people received two full doses. Eleanor Riley, professor of Immunology and Infectious Disease at the University of Edinburgh, said Oxford and AstraZeneca needed to answer questions about their results “clearly and completely.” “Trust is at a premium when it comes to vaccines and we must not do anything that might in any way undermine that trust,” she said. Full results are due to be published in medical journal The Lancet, though no date has been given. Pfizer and BioNTech said earlier this month that their vaccine is 95% effective, and Moderna said its product appears to be 94.5% effective, according to preliminary data. Unlike the Pfizer and Moderna vaccines, the Oxford-AstraZeneca jab does not need to be stored at freezer temperatures, making it potentially easier to distribute, especially in developing countries. It is also cheaper, because AstraZeneca has agreed not to profit from it during the pandemic. The British government and its scientific advisers have expressed confidence that multiple vaccines will win approval, but say all decisions are up to the regulator. “They will make an assessment with lots of data that is not currently public domain on efficacy and on safety,” said England’s Chief Medical Officer, Chris Whitty. “I think it’s always a mistake to make too many judgements early before we have the full information and particularly before the regulator, the independent regulator, has had their chance to look at the data and make an assessment.” from https://ift.tt/378VCHK Check out https://takiaisfobia.blogspot.com/ LONDON — AstraZeneca and Oxford University on Wednesday acknowledged a manufacturing error that is raising questions about preliminary results of their experimental COVID-19 vaccine. A statement describing the error came days after the company and the university described the shots as “highly effective” and made no mention of why some study participants didn’t receive as much vaccine in the first of two shots as expected. In a surprise, the group of volunteers that got a lower dose seemed to be much better protected than the volunteers who got two full doses. In the low-dose group, AstraZeneca said, the vaccine appeared to be 90% effective. In the group that got two full doses, the vaccine appeared to be 62% effective. Combined, the drugmakers said the vaccine appeared to be 70% effective. But the way in which the results were arrived at and reported by the companies has led to pointed questions from experts. The partial results announced Monday are from large ongoing studies in the U.K. and Brazil designed to determine the optimal dose of vaccine, as well as examine safety and effectiveness. Multiple combinations and doses were tried in the volunteers. They were compared to others who were given a meningitis vaccine or a saline shot. Did researchers mean to give a half dose?Before they begin their research, scientists spell out all the steps they are taking, and how they will analyze the results. Any deviation from that protocol can put the results in question. In a statement Wednesday, Oxford University said some of the vials used in the trial didn’t have the right concentration of vaccine so some volunteers got a half dose. The university said that it discussed the problem with regulators, and agreed to complete the late stage trial with two groups. The manufacturing problem has been corrected, according to the statement. What about the results themselves?Experts say the relatively small number of people in the low dose group makes it difficult to know if the effectiveness seen in the group is real or a statistical quirk. Some 2,741 people received a half dose of the vaccine followed by a full dose, AstraZeneca said. A total of 8,895 people received two full doses. Another factor: none of the people in the low-dose group were over 55 years old. Younger people tend to mount a stronger immune response than older people, so it could be that the youth of the participants in the low-dose group is why it looked more effective, not the size of the dose. Another point of confusion comes from a decision to pool results from two groups of participants who received different dosing levels to reach an average 70% effectiveness, said David Salisbury, and associate fellow of the global health program at the Chatham House think tank. “You’ve taken two studies for which different doses were used and come up with a composite that doesn’t represent either of the doses,″ he said of the figure. “I think many people are having trouble with that.″ Why would a smaller first dose be more effective?Oxford researchers say they aren’t certain and they are working to uncover the reason. Sarah Gilbert, one of the Oxford scientists leading the research, said the answer is probably related to providing exactly the right amount of vaccine to trigger the best immune response. “It’s the Goldilocks amount that you want, I think, not too little and not too much. Too much could give you a poor quality response as well,’’ she said. “So you want just the right amount and it’s a bit hit and miss when you’re trying to go quickly to get that perfect first time.” What are the next steps?Details of the trial results will be published in medical journals and provided to U.K. regulators so they can decide whether to authorize distribution of the vaccine. Those reports will include a detailed breakdown that includes demographic and other information about who got sick in each group, and give a more complete picture of how effective the vaccine is. Moncef Slaoui, who leads the U.S. coronavirus vaccine program Operation Warp Speed, said Tuesday in a call with reporters that U.S. officials are trying to determine what immune response the vaccine produced, and may decide to modify the AstraZeneca study in the U.S. to include a half dose. “But we want it to be based on data and science,” he said. from https://ift.tt/363JAQu Check out https://takiaisfobia.blogspot.com/ Hawaii Is Riding Out the COVID-19 Storm. But Geographic Isolation Isnt the Blessing it May Seem11/25/2020 When Hawaii’s coronavirus infections were rising in late August, Honolulu Mayor Kirk Caldwell visited doctors in the COVID-19 ward of The Queen’s Medical Center, the state’s largest hospital. “I could see it in their faces and in their eyes,” Caldwell says. “The concern and fear they had that, by the following week, if things didn’t change, they were not going to be able to care for people, that they were going to have to put them in tents outside.” It was a crisis that leaders in the 50th state hoped they would never face. Between March and May, when a stay-at-home order was in place, Hawaii was averaging only a couple of new cases every day. But as some restrictions were lifted in June, resulting in a patchwork of state and local rules, the numbers soon began inching up. By late July, Hawaii was metaphorically ablaze, with the bulk of cases centered on Oahu, home to two-thirds of the state’s population. The island logged 119 new cases on July 30; by mid-August, it was averaging over 200 a day. Four days after his hospital visit, with more than 350 people hospitalized for COVID-19 symptoms on his island, Caldwell issued a second lockdown order with the blessing of Hawaii’s governor, David Ige. “Our hospital administrators had informed us if we [didn’t] do something, that they would become overwhelmed,” says Ige. Three months later, it seems Hawaii’s response may have worked. As a new, deadly wave of coronavirus infections sweeps across the U.S., Hawaii is one of the only states in the country experiencing a relative reprieve. It reported 113 new cases on Nov. 23, according to data from Johns Hopkins University, slightly more than its weekly average of 106 daily new cases; only Vermont reported fewer cases that day. 
Hawaii, the 40th most populous state, has a low case count even when adjusting for population—as of Nov. 23, it has a weekly average of 7 cases per 100,000 residents, the lowest in the country (by comparison, the highest per-capita figures belong to North Dakota at 160 and Wyoming at 154). Infection levels in Hawaii have stayed relatively steady since mid-September, while nearly every other state has experienced an increase. It remains to be seen whether a slight uptick in recent days could be the start of a new upward trend, however. How has Hawaii so far avoided another major COVID-19 surge? The clearest reason is apparent on any world map. “Geography, it’s so obvious, yet it plays such a big role,” says Thomas Lee, an assistant professor of epidemiology at the University of Hawai‘i at Mānoa and co-chair of the Hawaiʻi Pandemic Applied Modeling Work Group; he also served as the lead outbreak modeler and forecaster for the Hawaii Emergency Management Agency this spring. For Hawaii, the Pacific Ocean has helped serve as the world’s biggest moat. Travelers arriving there by plane—essentially the only means of reaching the state, with cruise lines shuttered--have since March been required to self-quarantine for two weeks (though since mid-October, visitors can avoid isolation if they test negative within 72 hours of arrival). While the rules may have kept visitors from seeding new infection clusters, they have also devastated Hawaii’s tourism sector, which accounts for nearly a quarter of its economic activity. Some Hawaiian leaders still don’t think they’re doing enough to prevent infections—part of a broader split among Hawaiians over whether and how to welcome visitors back during a pandemic. Derek Kawakami, mayor of Kauai County, says new infections on his islands are related to the reopening of travel from the continental U.S. He’s proposed a requirement that travelers get tested a second time after arriving. “This virus requires layer upon layer of intervention and mitigating measures,” Kawakami says. “We have to be able to respond quickly and boldly, because it can spread like wildfire.” Kauai’s recent uptick is tiny in relative terms—on Nov. 20, it reported six cases, its second-highest one-day count so far. But Kawakami has good reason to be worried: Kauai, home to around 72,000 people, has just nine ICU beds and 14 ventilators. Isolated and with limited health care capacity, an uncontrolled outbreak could quickly spell disaster. “The holy grail is prevention, so that is what we put our blood, sweat and tears into here on Kauai,” Kawakami says. “We’re almost always on the red line.” A similar imperative applies across the entire state of 1.4 million people. With nowhere to turn if Hawaii’s health care system is overwhelmed, leaders there have little choice but to be proactive in preventing COVID-19 outbreaks. “We are geographically remote and so our health system is really vulnerable,” says Catherine Pirkle, an associate professor of health policy and management at the University of Hawai‘i at Mānoa. “There really is a strong motivating factor to try to keep those numbers under control.” Hawaii’s isolation, then, is a double-edged sword. Thousands of miles of ocean are a good tool to stop travelers from seeding new coronavirus clusters, but if infections spin out of control anyway, assistance could be a long time coming. “We have been from the very beginning focused on the science and public health recommendations because we are 2,500 miles away from any help,” says Ige, the governor. “We learned a long time ago that we have to be self-reliant, and that we have to act as a community.” Epidemiologists say it’s difficult to attribute Hawaii’s relative COVID-19 success to any one factor, isolation included. For one thing, cold weather, which is forcing many Americans indoors where the virus can spread more easily, is unheard of in most of Hawaii; Honolulu’s Thanksgiving Day forecast is 82 and sunny. Other government measures may also have played a part. Unused hotel rooms in Oahu have been repurposed to isolate COVID-19 patients, and restrictions on inter-island travel have prevented local spread. Mask-wearing has helped as well. On Kauai and Oahu, mask mandates were instituted in April, while an August survey found that 96% of Hawaiians are masking up. Unlike for many Americans, wearing masks to prevent illness isn’t an entirely foreign concept for Hawaiians; public health experts say that’s likely due to the state’s relative proximity to Asia, where facial coverings tend to be more prevalent. Many Hawaii residents who commented for this article also cited the state’s heritage and cultural values as a reason for high adherence to public health mandates. “We have a culture here that comes from the first peoples, the native Hawaiians,” Caldwell says. “The term Kuleana…means responsibility, and it does pervade the people here in Hawaii.” Still, leaders in Hawaii have come under pressure over COVID-19 restrictions. Caldwell says people sometimes drive down his street to yell outside his house, either at him or his wife and daughter. Some people certainly have reason to be upset—travel restrictions have ravaged the state’s tourism-dependent economy; Hawaii’s overall unemployment rate was more than 14% in October, according to the U.S. Department of Labor, the worst in the country for the second month in a row. Caldwell says he understands people’s frustrations, but needs to prioritize public health. “People are impacted. Their businesses are impacted. Their life dreams are impacted,” he says. “I get why they’re upset. But I also believe that in order to protect health and safety, which is always the most important thing, that comes first. And I think it’s tied directly to the economy, too.” After months of travel restrictions, Ige says the state’s pre-travel testing program is finally bringing in visitors and helping Hawaiians get back to work, while keeping COVID-19 levels stable. Still, despite Hawaii’s current low case counts, it remains to be seen if the state’s leaders can revive a tourism-based economy while at the same time staving off a devastating rise in cases—which would likely put the tourism business back on ice anyway. “We depend on travel,” Ige says. “But every day I get comments from members in our community that continuing to bring visitors into Hawaii during this time is really putting our community at risk…it’s something that weighs very heavily on myself and the mayors every day as we see the virus counts increasing on the mainland.” —With reporting from Emily Barone from https://ift.tt/2IZUCO9 Check out https://takiaisfobia.blogspot.com/ The Duchess of Sussex has revealed that she had a miscarriage in July, giving a personal account of the traumatic experience in hope of helping others. Meghan described the miscarriage in an opinion piece in the New York Times on Wednesday. She wrote: “I knew, as I clutched my firstborn child, that I was losing my second.” The former Meghan Markle and husband Prince Harry have an 18-month-old son, Archie. The duchess, 39, said she was sharing her story to help break the silence around an all-too-common tragedy. “Losing a child means carrying an almost unbearable grief, experienced by many but talked about by few,” she wrote. “In being invited to share our pain, together we take the first steps toward healing.” In a startlingly intimate account of her experience, the duchess described how tragedy struck on a “morning that began as ordinarily as any other day: Make breakfast. Feed the dogs. Take vitamins. Find that missing sock. Pick up the rogue crayon that rolled under the table. Throw my hair in a ponytail before getting my son from his crib. “After changing his diaper, I felt a sharp cramp. I dropped to the floor with him in my arms, humming a lullaby to keep us both calm, the cheerful tune a stark contrast to my sense that something was not right.” Later, she said, she “lay in a hospital bed, holding my husband’s hand. I felt the clamminess of his palm and kissed his knuckles, wet from both our tears. Staring at the cold white walls, my eyes glazed over. I tried to imagine how we’d heal.” Meghan, an American actress and star of TV legal drama “Suits,” married Harry, a grandson of Queen Elizabeth II, in a lavish ceremony at Windsor Castle in May 2018. Their son was born the following year. Early this year, the couple announced they were quitting royal duties and moving to North America, citing what they said was the unbearable intrusions and racist attitudes of the British media. They recently bought a house in Santa Barbara, California. from https://ift.tt/3m3QDON Check out https://takiaisfobia.blogspot.com/ With three vaccine makers saying that their COVID-19 vaccines are at least 90% effective, the U.S. government is gearing up plans to distribute the shots across the country. In a Nov. 24 briefing, members of the Department of Health and Human Services (HHS)—which oversees the Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration (FDA)—and Operation Warp Speed provided the latest information on the vaccine rollout. Most importantly, HHS Secretary Alex Azar said he’s ready to work with the incoming Biden administration’s transition team on vaccine rollout plans, following a decision by the General Services Administration to begin the handoff process. “I reiterated my firm commitment that the transition planning and execution will be professional, cooperative and collaborative in the best spirit of looking out for the health and wellbeing of the American people,” Azar said during the briefing. He previously said that he would not initiate transition communications until the GSA decision. Here’s what the Operation Warp Speed team, which is tasked with supporting development, manufacturing and distribution of COVID-19 vaccines, revealed during the briefing: An FDA committee will meet Dec. 10 to review Pfizer’s vaccineA panel of independent experts convened by the FDA will review studies from each vaccine to determine if they are safe and effective enough to distribute to the public. First up are Pfizer and German biotech firm BioNTech, which filed an emergency use authorization (EUA) request for their COVID-19 vaccine on Nov. 20. The FDA group will meet to evaluate the shot, which the companies say is 95% effective in protecting against COVID-19, on Dec. 10. Moderna is expected to submit a similar request to the FDA soon, and the committee will meet again to evaluate the Massachusetts-based company’s application. AstraZeneca will likely follow as well. The first batch will include 6.4 million dosesGen. Gus Perna, chief operating officer of Operation Warp Speed, said that he told state health departments on Nov. 20 that 6.4 million doses of vaccine will be available once the FDA issues the first EUA (at this point, Pfizer’s shots will most likely be the first to get the green light). “What will happen if more becomes available after the initial push is, we will go out following a weekly cadence to distribute [doses] accordingly, with 40 million [doses] by the end of the year.” How much vaccine each state initially receives will be based on a rough per capita calculation. Pfizer has conducted dry run shipping drillsPfizer’s and BioNTech’s vaccine, which is based on a new mRNA technology, needs to be kept at -70 C° to -80 C°. In the U.S., Pfizer has designed special thermal packaging to ship its doses. If replenished with dry ice, the doses can remain in those containers for 15 to 20 days. Once removed, the vials can be stored in a refrigerator for up to five days. In order to train workers who are receiving the doses, Pfizer has conducted dry runs of shipping its vaccine to 10 locations. Perna said such trials would be done with all 64 public health jurisdictions receiving vaccines from the CDC—most of which are state health departments—to make sure those administering the vaccine are comfortable handling it. “I’ve personally gone through the process, put my hand in the box, pulled out trays, and timed myself in the execution, and it’s a very, very doable process,” he said. The Pfizer containers will include 975 doses. “We feel confident that if states plan dosages in the right locations, with the drumbeat and cadence of [regular manufacturing], administration of the vaccine to a minimum amount of 975 doses is very doable within 20 days,” he added. Once the first batch of vaccines are shipped, the next batch will be dispatched within 21 days for the Pfizer vaccine and 28 days for the Moderna vaccine, so people who received their first dose will be able to get their second shot. Once the manufacturing reaches a regular cadence, shipments will be distributed weekly. States will be able to order vaccines through a 24/7 system, and the doses will be shipped the following day. It will take months until most of the American population can be vaccinatedVaccine manufacturing will gradually increase as more shots are authorized by the FDA, and people will be vaccinated according to guidelines set by the CDC’s Advisory Committee on Immunization Practices (ACIP), among others. The ACIP recently released principles that it says should guide who gets vaccinated first; these factors include risk as well as equity. Health care workers, first responders and others with essential jobs will be first in line, with the elderly and those with chronic health conditions next. It likely won’t be until next spring or summer that the majority of the American public will be eligible to get vaccinated. “We are bridging to when we have vaccines first for the most vulnerable like health care workers, first responders and eventually the broad American population,” Azar said. “[Until then] wash your hands, watch your distance and wear face coverings when you can’t watch your distance, and stay out of settings where you can’t do those things.” from https://ift.tt/377wctQ Check out https://takiaisfobia.blogspot.com/ Public-health officials have for weeks been urging Americans to avoid gathering for Thanksgiving this year, lest the holiday turn into a national super-spreading event. But for those who still plan on getting together, experts say an outdoor get-together is far safer than prolonged time indoors, where it’s easier for the virus that causes COVID-19 to spread. Whether an outdoor Thanksgiving is really feasible, however, largely depends on, well, the weather. Good news: a look at the Thanksgiving Day forecast reveals that, for most of the U.S., temperatures will be above normal and the skies will be clear. “When it comes to Thanksgiving weather, most of the country will have lucked out,” says John Homenuk, a meteorologist at New York Metro Weather, via email. “Warmer than normal air and calm conditions are expected from the Plains states to the Great Lakes and Tennessee Valley. Cooler, but mostly pleasant weather is also expected in the Pacific Northwest down the West Coast to the Desert Southwest. Outdoor activities should at least be possible in many of these regions, but especially the Central U.S. and Ohio Valley, where temperatures are expected to be several degrees warmer than normal.” Homenuk also sent along this map, which shows expected temperatures across the U.S. in terms of how far above or below normal they’re expected to be for this time of year: 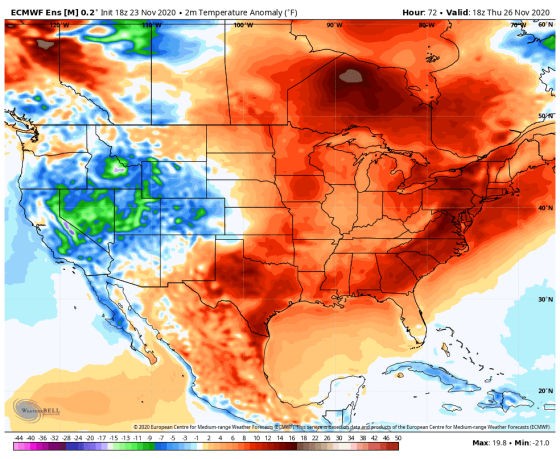
However, an outdoor Thanksgiving may be less of an option across much of the U.S. East Coast, which is likely in for some wet weather. “Areas like Philadelphia, New York City and Boston are likely to be dealing with showers and dreary conditions throughout the morning and early afternoon,” says Homenuk. “Some clearing is possible later in the day if a cold front can sweep through quickly enough.” 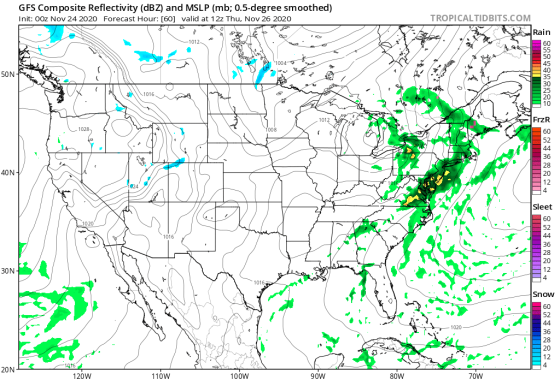
Even with this weather forecast in mind, it’s still safer to avoid gathering this year with anybody you don’t already live with (and no, getting a single COVID-19 test can’t guarantee a safe Thanksgiving). But if you’re committed to having people over or heading somewhere else despite the risks, it’s looking like turkey al fresco will be a viable option for many. Still, it’s wise to keep the guest list as short as possible and ensure everybody stays masked and distanced as much as possible; it’s especially important to keep people apart while they’re masks-down and eating. One other suggestion: rather than inviting a bunch of people over for an all-day hang and dinner, have a smaller group over for a distanced outdoor hangout for part of the day, then do dinner with your immediate household later. from https://ift.tt/3m2mAa6 Check out https://takiaisfobia.blogspot.com/ In February 2019, the Kaiser Permanente health system announced a new kind of medical school. The school would be built “from the ground up” to prepare students for the complexities of the U.S. medical system. The curriculum would emphasize cultural competency, patient and provider well-being, and the elimination of socioeconomic disparities in the medical system. Students would see patients right away, and hands-on learning would replace many lectures. What’s more, the first five graduating classes would pay nothing to attend; Kaiser hoped this would attract a student body more diverse than the typical U.S. medical school. “The school will help shape the future of medical education,” promised Kaiser CEO Bernard Tyson, who died unexpectedly, reportedly of a heart attack, about nine months after the announcement. That future felt a good deal more urgent by the time the Kaiser Permanente Bernard J. Tyson School of Medicine opened its doors in Pasadena, Calif., in July. The COVID-19 pandemic had put a hold on almost every facet of “normal” life, and the medical system was scrambling to treat millions of patients with a new and terrifying disease, a disproportionate number of them Black and brown. The streets were filled with people protesting police brutality and racism, as a nation that had long overslept awoke to the disparities woven into almost every American institution. “Our country doesn’t just have a pandemic; it also has a renewed recognition of centuries of racism,” says Kaiser’s founding dean Dr. Mark Schuster. “We need to make sure that our students understand our history.” Kaiser isn’t alone there, of course. Medical schools all over the world have had to adjust on the fly this year, in ways both practical and ideological. First, schools had to figure out how to remotely train students in skills taught hands-on before lockdowns. Then, in the U.S., schools were also forced to grapple with their roles in a health care system that often fails to keep Black and brown patients well. That meant learning how to produce doctors who could help chip away at those disparities moving forward. With no warning and no instruction manual, medical schools are figuring out how to train a generation of post-pandemic doctors for a world still taking shape. The foundations of the American medical education system haven’t changed much for decades. The first two years are a mad rush to attend lectures and memorize as much information as humanly possible, since students usually take the first part of their medical licensing exam after their second year of school. In their third year, students start clinical rotations in hospitals, then spend most of their fourth trying to find a match for their next phase of training: medical residency. The coronavirus pandemic upended all of that this spring. Classes could no longer happen in person, let alone in large lecture halls. Students couldn’t go to hospitals for training, since facilities needed to conserve resources and personal protective equipment. And travel restrictions made it difficult for fourth-year students to do “audition rotations” at hospitals where they hoped to complete residency. Fiona Chen, who was in her third year at Brown University’s Warren Alpert Medical School this past spring, went from spending around 40 hours a week in the clinic to watching one Zoom lecture a week and volunteering for a coronavirus information hotline. “We basically put a pause on our entire lives,” she says. They couldn’t stay paused forever. Schools had no choice but to adapt, which, for many, opened the door to overdue changes—changes that are coming in handy with COVID-19 again surging and new lockdowns being enacted. “A lot of the inertia and conventions of medicine are being broken down,” says Dr. William Jeffries, vice dean for medical education at the Geisinger Commonwealth School of Medicine in Pennsylvania. “Advances in medical education are now happening at light speed.” Though some students returned to the classroom later in the year, step one this spring was bringing traditional classes online—a fairly easy task for most schools in the developed world, though less so for schools in places like Southeast Asia and Africa, where Internet access is spottier. In developed nations, at least, the shift enabled schools to look critically at the way they were teaching before the pandemic. Kaiser’s preexisting plan to teach students anatomy using virtual reality simulators, rather than cadavers, proved fortuitous. Imperial College London gave students access to a video library of old patient interviews and exams. At New York University’s Grossman School of Medicine, professors began prerecording their lectures so students could watch in advance and use class time for livelier discussion. “Lectures have been fading as a useful didactic model for 10 years, but we continue to use them,” says Dr. Steven Abramson, NYU’s vice dean for education, faculty and academic affairs. The pandemic may finally catalyze lasting change. When third- and fourth-year students were yanked from hospitals this spring, many schools pivoted to telemedicine appointments. (This wasn’t unique to medical schools; remote visits surged across the health care system.) After the new academic year started this summer, third-year students at Geisinger spent the first 10 weeks learning how to take patient assessments and develop treatment plans over Zoom. “When clinical care changes, medical student education follows,” says Dr. Alison Whelan, chief medical education officer at the Association of American Medical Colleges (AAMC). That required teaching students “webside” (as opposed to bedside) manner, to prepare them for a clinical practice likely to be far more virtual than that of their predecessors. “If you’re not shaking hands, how do you make that initial connection [with a patient]?” Whelan asks. Students have also been honing the skills needed to perform the behind-the-scenes work that goes into a telemedicine appointment—like how to handle patient privacy when a spouse wanders into the room, or what to do when a patient can’t figure out how to work the web platform, Whelan says. Still, you can’t take an EKG or draw blood virtually. To continue teaching skills like these when students were sent home, Geisinger built an “e-ICU” that allows students to see what’s going on in hospital rooms, and remotely do the sort of trainee doctor work they’d have done in person before the pandemic. Through a webcam, they can ask resident doctors on duty to perform certain exams or tests, as if they’re actually at the patient’s bedside, and then get immediate feedback from the resident. The model worked so well that Geisinger plans to continue the e-ICU and the school’s broader telemedicine training even as students return to regular clinical work, Jeffries says. Doctors who are digitally literate and comfortable using telemedicine could help expand access to care in the future, he says. Programs like the e-ICU could also help connect doctors in small community clinics with specialists who may not be available locally. “I come from a small town in the middle of nowhere. We don’t even have a post office,” says Dr. Cass Lippold, a critical-care fellow at Geisinger who oversaw the e-ICU program. “This will be great to help those people who don’t have access to a hospital.” Programs like these could also improve doctors’ work-life balance. “If you’re a physician with a couple young kids at home, telemedicine has really opened an opportunity to work from home a couple days a week and still see patients,” says NYU’s Abramson. Jeffries notes that moving classes online could also make it easier for prospective doctors with physical or learning disabilities to participate, since they could tailor their environment to fit their needs. 
The shift to online learning was a logistical undertaking, but the harder work may be producing doctors who are better equipped to take on the systemic issues exposed by the pandemic, like race-based health disparities, uneven access to care and ballooning treatment costs. At Kaiser Permanente, that preparation began before students even started classes this summer. The entire class was invited to a virtual check-in to discuss the racial-justice movement, and the conversation hasn’t stopped since, says 26-year-old first-year student Cruz Riley, who has a special interest in Black maternal health. “You would think we would be talking about what we watched on Netflix,” he says. “But we are always talking about systematic inequality, and we are always bouncing ideas off each other.” It helps that Kaiser integrated lessons about inequality and social determinants of health throughout its entire curriculum, a move that many older schools are rapidly trying to replicate. Kaiser also requires first-year students to take a class on mental health and overcoming stress, and to visit an on-campus psychologist three times during their first semester. Those services, available free of charge throughout their medical education, are part of a program Kaiser implemented to counter high rates of burnout and mental distress among medical students: studies estimate more than 25% worldwide show signs of depression, and about 10% of suicidal thinking. But it has also provided valuable support as students of color do the emotional labor of living through constant reminders of racism in America, says 25-year-old first-year student Emilia Zevallos-Roberts, who was born in Ecuador. Courses on health disparities and racism in medicine aren’t new in the U.S., but they also haven’t been terribly effective. Racism is still a problem in medical schools, as well as the wider medical system. A study from earlier this year found that about 25% of students who identify as Black, Hispanic/Latinx or American Indian/Alaska Native experienced race-based discrimination during medical education. That doesn’t stop after graduation. “There were so many comments that I had to endure in my undergraduate years, in my medical school years,” Dr. Tsion Firew, an emergency-medicine physician at New York City’s Columbia University, who is Black, told TIME this summer. “When I walk into my hospital, it’s not [diverse] like New York City. The second you walk into the hospital, you are reminded that you’re not part of the majority.” Medicine and medical education remain very white fields in America. In 2019, out of nearly 38,500 medical school professors in the U.S., 755 (2%) identified as Black, around 1,000 (2.6%) identified as Hispanic or Latino, and just 37 (0.01%) identified as American Indian or Alaska Native, according to AAMC data. More than 29,000, or 75%, identified as white. For context, about 60% of the total U.S. population identifies as white, while about 12% identify as Black, 18% as Hispanic, 5.6% as Asian and less than 1% as American Indian/Alaska Native, according to data from the U.S. Census Bureau’s American Community Survey. Given that dynamic, it’s not hard to understand why many schools haven’t historically done a good job teaching concepts like cultural competency (the ability to connect with and treat patients from all backgrounds) and social determinants of health (the myriad ways socioeconomic factors affect a person’s well-being). Many also fail to correct (and in some cases even perpetuate) racist and incorrect stereotypes about biological differences between Black and white patients. One 2016 study found that, out of about 400 medical students and residents surveyed in the U.S., half held false beliefs, such as that Black people have a higher pain tolerance or physically thicker skin than white people. If students are steeped in these incorrect stereotypes, rather than very real social determinants of health, they may contribute to a system of racially insensitive, and potentially harmful, medical care. Many schools were already working to fix that before the pandemic, but mainstream conversations about inequality and racism have hastened the process. Chen, currently a fourth-year student at Brown, says she’s noticed that race and social factors now come up when discussing every patient case, whereas before they were often relegated to stand-alone lectures or lessons. Tian Mauer, a third-year student at Geisinger, has noticed the same thing. And for schools across the U.S., the AAMC has guidelines for teaching equity, diversity and inclusion in medicine. “COVID has really highlighted for some for whom it had not yet clicked that the social determinants of health are really critical,” Whelan says. Of course, it will take more than a few lectures to address centuries-old disparities in medical care, particularly because systemic racism has so many tendrils. It’s not enough to train physicians on implicit bias and cultural sensitivity when Black and Hispanic Americans’ health suffers due to poverty and segregation built up over centuries—or when many people from these communities can’t afford to become doctors themselves, perpetuating cycles of mistrust in a heavily white medical system. At most medical schools, the student body looks a lot like the faculty. Together, Black and Hispanic students made up less than 15% of the national medical student population during the 2019–2020 school year, AAMC data show. People who identified as American Indian/Alaska Native made up just 0.2%. Wealth disparities go a long way to explaining why: medical school tuition and fees can easily top $60,000 per year, and the average new doctor graduates with about $200,000 in debt, according to AAMC data. Before the pandemic, a small but growing group of schools were trying out a way to fix that: offering free or heavily discounted tuition. NYU permanently waived its $55,000 annual tuition in 2018. Geisinger now offers free tuition for students who agree to practice within its health system. Kaiser’s free tuition offer will go to its first five graduating classes. The pandemic may accelerate conversations about affordability, especially as financial stress stretches on. Dr. Steven Scheinman, the dean at Geisinger, says a stronger reliance on remote learning could push the school’s tuition down over time. NYU and about a dozen other U.S. medical schools are also part of a consortium studying how an accelerated medical school schedule—three years instead of four—affects learning, student finances and licensing and placement for new doctors. Cutting a year of school would get doctors out into the field faster, saving them a year of expenses. More than a dozen U.S. medical schools, including NYU and each of the four medical schools in Massachusetts, along with many in the U.K., like the University of Cambridge and Oxford University, allowed their students to graduate early this spring to help with pandemic response. In a worst-case scenario, this fall’s spike could necessitate something similar. The U.S. medical school system also has missed opportunities presented by COVID-19. For example, relatively few schools changed their admission requirements in ways that could have made life easier for applicants. To apply for most U.S. medical schools, students still had to take a $320 hours-long standardized exam called the Medical College Admission Test (MCAT). A grassroots group called Students for Ethical Admissions called on schools to waive that requirement, citing the risks of disease spread that come with sitting for hours in an exam room with strangers, but only a handful of schools, including Stanford and the University of Minnesota, did so. The AAMC, which administers the test, maintains that all students should still take the MCAT. And not all schools have used the moment to update their curricula, nor done a seamless job of bringing learning online. A study of U.K. medical students found that the majority experienced some disruption to their normal training. “This is a detriment to my education, sitting in my bedroom trying to focus when my parents are home working,” agrees 23-year-old Elli Warsh, who is in a nursing program at New Jersey’s Rutgers University. Warsh and her classmates were pulled out of the hospital from March to July and didn’t see any patients for those months. They had to practice skills like full-body assessments on family members or roommates; some students who lived alone used teddy bears. Now, Warsh says, she has no idea if her skills will be on par with previous new nurses when she graduates in May. Those are real fears, particularly for students who aren’t attending big-name, richly endowed medical schools that were able to adjust on the fly, and for students shouldering burdens like financial distress and childcare during the pandemic. Time will tell how they fare when their residency placements come around. In the meantime, students like Zevallos-Roberts, from Kaiser’s School of Medicine, find optimism in the disruption. “Although the pandemic is obviously devastating,” Zevallos-Roberts says, “I’m hoping that the energy and momentum for change that we’re seeing now, that we’re able to bring that forward when we’re graduating three years from now.” from https://ift.tt/2KCSGeM Check out https://takiaisfobia.blogspot.com/ |
Authorhttps://takiaisfobia.blogspot.com/ Archives
April 2023
Categories |
 RSS Feed
RSS Feed
