|
Disposable e-cigarettes like Puff Bar are now far more popular among teenagers than the pod-based products from once-dominant brand Juul, according to new data from the U.S. Centers for Disease Control and Prevention (CDC) and Food and Drug Administration (FDA). Each year, the CDC and FDA survey thousands of U.S. middle and high school students about their use of tobacco products, including e-cigarettes—which were meant to give adult smokers a less-dangerous alternative to cigarettes, but have in recent years become popular among underage users. The 2021 edition was conducted entirely during the pandemic, meaning some teenagers took the survey at home, instead of in their classrooms as usual. As a result, the report’s authors caution that it shouldn’t be directly compared to previous surveys. Even with that caveat, the report shows clear shifts in teenage vaping habits during the pandemic. About 11% of high schoolers and 3% of middle schoolers said they had vaped in the 30 days prior to taking the survey—far below the 20% and 5% who said so last year, respectively. That’s likely in part because remote learning affected students’ access to e-cigarettes, according to the report’s authors, so there’s no guarantee the downward trend will continue in the future. Teen e-cigarette preferences are also changing dramatically. For years, Juul—a company that makes flash-drive-like devices that run on prefilled cartridges of nicotine e-liquids—was thought to be the most popular brand among teens, and has drawn criticism for marketing in ways that appeal to teenagers. (The company has repeatedly denied that it ever purposely targeted teenagers.) But in the new report, more than half of students who vaped said they used disposable e-cigarettes, while about 29% said they used pod-based products. About 85% of youth vapers used products in flavors including fruit, candy and mint. Less than 6% of high school vapers said Juul was their preferred brand, compared to 26% who used products from Puff Bar—a brand that makes disposable e-cigarettes in flavors like Blue Razz and Watermelon. The FDA tried to remove Puff Bar from the market in 2020 for violating regulatory requirements, but it reemerged this year using a type of lab-made nicotine that some in the vaping industry argue the FDA can’t regulate. Puff Bar did not respond to TIME’s request for comment. Disposable products, in general, are not regulated as tightly as pod-based products like Juul’s, which may explain some of the shift in teen preferences. At the beginning of 2020, the FDA forbade the sale of all flavored pods that had not received agency approval, in hopes of easing the teen vaping trend; that’s why brands like Juul now only sell menthol- and tobacco-flavored liquids. The ban was controversial, since many adult e-cigarette users also prefer flavored products. It did not extend to disposable vapes, hence why they’re still available in a range of flavors. That may not be true for long, though. Last September, all U.S. e-cigarette makers were supposed to submit applications proving that their products were positive for public health—that they could be beneficial enough for adult smokers to outweigh their potential health risks or popularity among teenagers and recreational users. So far, the FDA has denied more than a million applications for flavored e-cigarettes, meaning they can no longer be legally sold in the U.S. It is still reviewing applications from big-name brands like Juul and Vuse, the e-cigarette made by tobacco company R.J. Reynolds. Juul’s application, in particular, has drawn condemnation from many public-health groups, which have urged the FDA to reject it due to the company’s role in the teen vaping crisis. Juul may have helped spark that problem—but as the new federal data show, that spark has largely burned out. from https://ift.tt/2Y24qhI Check out https://takiaisfobia.blogspot.com/
0 Comments
There’s been quite a bit of news about COVID-19 booster shots lately, and it’s been more confusing than reassuring. Public health officials don’t agree on whether everyone needs a booster yet, and in the same way that the vaccines were rolled out to different groups of people one category at a time as government agencies reviewed studies to make sure they were safe and effective, boosters are being doled out to certain people first. Here’s where we stand for now. If the vaccines work, why do I need a booster?The three vaccines that are currently available in the U.S.—from Pfizer-BioNTech, Moderna and Johnson&Johnson-Janssen—are still highly effective in protecting you from getting COVID-19 disease. They are slightly less effective in protecting you from getting infected with the virus in the first place, but people who are vaccinated still have a five times lower rate of infection with SARS-CoV-2 than people who aren’t vaccinated, and a more than 10-fold rate of hospitalizations and deaths from the Delta variant of the virus. Several months after getting the vaccine, the level of antibodies against the COVID-19 virus starts to wane, so public health officials feel that it makes sense to boost those numbers back up with another dose of the shot. There is some evidence that with each additional shot, the body starts making higher quality antibodies that are better at recognizing and blocking new variants of the virus, including Delta. Who is eligible for a booster shot right now?In August, the U.S. Food and Drug Administration (FDA) and Centers for Disease Control (CDC) authorized booster shots for anyone who has been vaccinated with either Pfizer-BioNTech or Moderna shots against COVID-19 and has a compromised immune system due to, for example, chronic illnesses, transplant surgery or cancer treatment, among others. As of September 2021, that recommendation expanded to include certain people who received the Pfizer-BioNTech vaccine. The FDA and CDC now recommend that people who were originally vaccinated with two doses of the Pfizer-BioNTech shot should get a booster six months after their last dose if:
The FDA and CDC also included others who initially got the Pfizer-BioNTech vaccine, but said the booster wasn’t mandatory for them. The following people can get a booster if they want to:
These decisions came after the FDA and CDC reviewed studies from Pfizer-BioNTech on the safety and efficacy of a booster dose among a broader group of people who received the approved two doses of the shot, and then got another dose at least six months later. The agencies also looked at data from Israel, where some people have been getting booster doses since July 30. Taken together, these data showed that after several months, levels of virus-fighting antibodies start to drop—though they also showed that people are still protected from getting severe COVID-19 disease and getting hospitalized. Still, the waning immunity worries public health officials, especially since the Delta variant, which spreads more quickly, is now the dominant version of the virus behind new infections around the world. In a study of a several hundred people who received a booster dose, researchers from Pfizer-BioNTech reported that the additional dose is safe and can raise antibody levels back up to those achieved immediately after the second dose, particularly among people over age 65 years. Israeli health officials also reported that older individuals who got a booster dose were 11 times less likely to get infected with SARS-CoV-2, and 19.5 times less likely to get severe COVID-19 than those receiving only two doses. If I’ve been vaccinated with Moderna or Johnson&Johnson-Janssen, can I get a booster?At the moment, no. The FDA and CDC have only reviewed the safety and efficacy of boosters using the Pfizer-BioNTech vaccine. Moderna has submitted its data to the FDA to review, and the agency is expected to make a recommendation in coming weeks. Johnson&Johnson-Janssen, which makes the only authorized single-shot COVID-19 vaccine in the U.S., has not yet provided the FDA with data on its booster, although the companies have reported early data showing that a second shot can increase antibody levels by nine-fold in people ages 18 to 55 years compared to after the initial single dose. Why are decisions about Moderna and Johnson&Johnson-Janssen boosters taking longer?The timeline for the booster doses is mirroring that of the original vaccines. Pfizer-BioNTech were the first to submit a request to the FDA for authorization of its vaccine. in November 2020; Moderna followed about 10 days later, and J&J filed its request in February 2021. The FDA issued emergency use authorization for all of them, based on two-month follow up data showing that the shots were both safe and efficacious, on a similar timeline, with Pfizer-BioNTech getting authorization first, followed by Moderna and then J&J. (In August 2021, after reviewing longer term data on safety and efficacy that stretched to six months, the FDA fully approved the two-dose regimen of the Pfizer-BioNTech vaccine, making it the first approved COVID-19 shot. Moderna has also provided longer follow up data to the FDA and is waiting for the agency’s decision about full approval.) In order to get authorization for a booster, each of the vaccine manufacturers had to conduct additional studies on the safety and efficacy of an additional dose, and then follow the people in those studies for several months to document any side effects as well as effects on immunity. Is the Pfizer-BioNTech booster a different vaccine from the original one?No, the booster shot of the Pfizer-BioNTech vaccine is the same dose, and the same vaccine used in the first two doses. Will the Moderna booster follow Pfizer-BioNTech’s model and be another dose of the same Moderna vaccine?It’s not clear yet, as Moderna is testing different versions of a booster. The company is testing a booster that is half the dose of the original two doses, one that is the same dose, another that is a different vaccine that targets the Beta variant, and finally one that combines the original and Beta variant boosters in a single shot. Moderna and the FDA will decide, based on the results of those studies, which booster will help the most people to stay protected against COVID-19. I’ve heard that people who received Moderna or Johnson&Johnson-Janssen are getting booster shots.This may be possible, but health officials say it’s not recommended. Researchers are still studying how people react to mixing doses from different vaccine manufacturers, so it’s not clear yet whether that’s a safe thing to do. And just as importantly, there are no data showing that getting a booster of a different vaccine will continue to protect you from COVID-19. Will I have to provide proof that I was originally vaccinated with the Pfizer-BioNTech shot in order to get a booster?Technically, yes, but CDC director Dr. Rochelle Walensky said that the CDC is urging vaccine providers not to deny people shots, so vaccinators likely would not be asking for documentation, opting instead to ask people to self-attest that they are eligible. However, vaccination sites will have different policies to verify eligibility. Walgreens, for example, says they will ask people to attest that they meet the requirements for a booster when they schedule their appointment, and again just before they get their shot, but pharmacists won’t require documentation. “Ultimately we are focused on removing barriers and improving access to vaccines,” a spokesperson tells TIME. On the other hand, according to a spokesperson for the UCLA Medical Center, vaccinators at the Los Angeles hospital are asking people to bring their vaccination card to verify that they originally received the Pfizer-BioNTech shot. For people who misplaced their card, providers are likely asking people to self-attest that they received the Pfizer-BioNTech shot, and that it’s been at least six months since their last dose. In general, sites probably won’t be asking for proof that you meet the age, underlying health condition, or occupational risk criteria. from https://ift.tt/3CUR4CM Check out https://takiaisfobia.blogspot.com/ When health experts talk about the remarkable efficacy of COVID-19 vaccines, they typically point to their ability to prevent severe disease and death. Fully vaccinated people can still get “breakthrough” infections from the virus that causes COVID-19—but compared to an unvaccinated person, they’re more than 10 times less likely to be hospitalized or die from their illness, according to U.S. Centers for Disease Control and Prevention (CDC) research. Officials often point to these impressive figures as evidence that we can tame COVID-19 into a mostly mild illness that behaves like a routine cold or flu, and thus with which we can coexist. After all, the vaccines were not designed to quash viral spread entirely; they were designed to defang the virus by preventing its worst outcomes. But people like April Zaleski know COVID-19’s worst outcomes aren’t limited to severe disease and death, even for the fully vaccinated. Zaleski, a 32-year-old from Idaho, caught COVID-19 in July 2021 after being vaccinated in January. She recovered after a couple weeks and thought the worst was behind her. But then her fatigue came back with force, along with brain fog, shortness of breath, vertigo and a skyrocketing heart rate. She began to suspect she had Long COVID, the name adopted by people who suffer symptoms long after their initial infection. Long COVID “was on my radar,” Zaleski says, “but having been vaccinated, I hoped that my chances of that happening were slim to none.” Indeed, the vast majority of vaccinated people who catch COVID-19 will not develop Long COVID, just as they will not die or go to the hospital; many won’t even have symptoms or realize they’re infected. But some will develop symptoms that don’t go away. Some, like Zaleski, already have. “There are so many people who are recovering from COVID, and that’s absolutely wonderful,” she says. But others “are struggling with long-term symptoms, and I just wish that people knew that side of COVID better.” Read more: Black Women Are Fighting to Be Recognized as Long COVID Patients In the pre-vaccine era of the pandemic, experts estimated that somewhere between 10% and 30% of people who caught COVID-19 had lingering symptoms of some kind and severity. Though it’s not entirely clear why that happens, the two leading theories are that the virus can trigger an autoimmune response that essentially makes the body attack itself, or that viral remnants sometimes linger in the body and cause lasting symptoms, says Yale University immunobiologist Akiko Iwasaki. Vaccines have changed the game somewhat. A vaccinated person is far less likely than an unvaccinated person to get infected, according to the CDC. The agency tracked COVID-19 cases in 13 U.S. states and/or major cities from April to mid-July 2021, and found that just 8% of them occurred among fully vaccinated people. That’s good news as far as long COVID is concerned: if you don’t get infected, you can’t develop Long COVID. But what about the unlucky people who do experience breakthrough infections? Research is just beginning to address that question. One study, published in the New England Journal of Medicine in July, analyzed 39 fully vaccinated Israeli health care workers who had breakthrough infections. Almost 20% of them still had symptoms six weeks later. Though its sample size was small, the study demonstrated that Long COVID is possible after a breakthrough infection. Another study, published in the Lancet Infectious Diseases in September, used self-reported symptom data from U.K. adults who tested positive for COVID-19 after being fully or partially vaccinated, as well as data from a control group of unvaccinated people who tested positive for the virus. They found that a fully vaccinated person who experienced a breakthrough infection was half as likely to have COVID-19 symptoms at least a month after diagnosis, compared to an unvaccinated person. That’s encouraging—but, again, the research showed that some vaccinated people are developing Long COVID. Dr. Megan Ranney, who co-leads Brown University School of Public Health’s Long COVID research initiative, estimates that somewhere around 5% to 10% of fully vaccinated people who get infected may develop lingering symptoms. But, she cautions, that’s based on a very small amount of preliminary data, so it’s hard to say for sure. It’s not even clear how many vaccinated people in the U.S. have been infected—which makes it pretty hard to tell how many developed Long COVID—since the CDC stopped tracking mild breakthrough cases this spring. Ranney says the risk of developing Long COVID is likely small enough that vaccinated people don’t need to be overly concerned. Everyone should take steps to reduce their potential exposure to the virus for many reasons, she says, but “Long COVID should not be a major driver of your decisions about what to do on a daily basis, if you are fully vaccinated.” Nevertheless, the risk is not zero. Lauren Nichols, vice president of the Long COVID support group Body Politic—a national organization that claims some 11,000 members—says her group hears from about five fully vaccinated prospective members each week. Anna Kern, a 33-year-old from the Detroit area, is one. She caught COVID-19 in April 2021 after being vaccinated months prior, and still has fatigue, difficulty walking or standing and a racing heart rate. “When I got COVID, there were very few people [who had gotten long COVID from a breakthrough infection],” she says. “There are more of us now, just by sheer numbers and time having passed.” Groups like Body Politic are fighting for their recognition. “COVID isn’t just about the mortality rate,” says Nichols, 33, who developed a case of Long COVID after catching the virus in March 2020 and is still sick. “It’s [also] about the…disability and chronic illness that occurs from it.” Nichols says that aspect of the pandemic is often ignored by public health authorities, who tend to talk about COVID-19 as a binary: either you develop a severe case that can be fatal, or you get a mild case and you’re fine. Long COVID doesn’t fit neatly into either box, but that doesn’t mean it should be ignored, Nichols argues. “Deaths and severe disease and hospitalization are…understandably what we are focused on. But Long COVID is an absolutely debilitating disease,” Iwasaki agrees. “It’s really been ignored to a large extent, and I really don’t understand why.” She says better tracking of breakthrough infections, as well as follow-up surveys that track patients’ symptoms over time (like those used in the recent Lancet study), could help increase understanding of Long COVID and the risk it poses to U.S. public health. Without that understanding, people who develop chronic issues after a breakthrough infection, like Zaleski and Kern, may be overlooked and left out of crucial efforts to develop treatments. The National Institutes of Health has earmarked more than a billion dollars for Long COVID research—but for it to be most effective, it needs to include a diverse, representative group of people. Read more: How COVID-19 Long Haulers Could Change the U.S. Disability Benefits System Nichols also says health officials should look to the past, drawing on the lived experience of people who developed other post-infectious chronic illnesses, like myalgic encephalomyelitis/chronic fatigue syndrome and chronic Lyme disease, to understand how to support Long COVID patients. Better access to specialized treatment centers; funding for patient-led advocacy and research groups; and collaboration with the entire chronic-disease community would all be good places to start, she says. And in the meantime, Nichols says public health agencies need to communicate that Long COVID is a possibility, both for unvaccinated and vaccinated people. As for how to minimize that risk, Ranney says any policy that slows the transmission of COVID-19—such as vaccine and mask mandates or restrictions on large indoor gatherings—will be doubly effective. Not only will it prevent acute cases of the virus, but it will also naturally make Long COVID less of a risk since fewer people are getting infected. “From a policy level,” she says, “the goal has to be reducing transmission. Period.” from https://ift.tt/3D3g9vk Check out https://takiaisfobia.blogspot.com/ When I kicked cocaine and heroin in 1988, I was told that there was only one way to get better: abstain forever from psychoactive substances including alcohol—and practice the 12 steps made famous by Alcoholics Anonymous. The only alternative, counselors and group members said, was “jails, institutions or death.” My addiction was so extreme that by the end, I was injecting dozens of times a day. So I grabbed the lifeline I was thrown and attended the traditional 12-step rehab program recommended by the hospital where I underwent withdrawal. But once I began to study the scientific data on addiction, I learned that these claims were not accurate. In fact, research shows that most people who meet full diagnostic criteria for having an addiction to alcohol or other drugs recover without any treatment or self-help groups—and many do so not by quitting entirely, but by moderating their use so that it no longer interferes with their productivity or relationships. There is no “one true way” to end addiction—and the idea that “one size fits all” can be harmful and even deadly in some cases. Until we recognize this and celebrate the variety of recovery experiences, September’s National Recovery Month and similar efforts to promote healing will fail to reach millions of people who could benefit. During an overdose crisis that killed more than 90,000 people in 2020 alone, a better understanding of how people really do overcome addiction is essential. Unfortunately, rehab hasn’t improved much since I attended in the late 20th century. At least two-thirds of American addiction treatment programs still focus on teaching the 12 steps and promoting lifelong abstinence and meeting attendance as the only way to recover. (The steps themselves include admitting powerlessness over the problem, finding a higher power, making amends for wrongs done, trying to improve “character defects” and prayer—a moral program unlike anything else in medicine.) Moreover, despite the fact that the only treatment that is proven to cut the death rate from opioid addiction by 50% or more is long-term use of either methadone or buprenorphine, only about one-third of residential programs even permit these effective medicines, and around half of outpatient facilities use them, typically short-term. Worse: when they do allow medication, most treatment centers also push people with opioid use disorder to attend the 12-step program, Narcotics Anonymous. That creates what can be deadly pressure to stop the meds. The group’s official literature says that people on methadone or buprenorphine are not “clean” and have only substituted one addiction for another. I have been contacted by more than one family who lost a loved one to overdose because their relative had rejected or prematurely ended medication based on this view. If we don’t start to view recovery more inclusively, we are denying hope and healing to those who benefit from approaches other than the steps. So, what does a more accurate and expansive view of recovery look like? To me, one of the most helpful definitions was devised by a group known as the Chicago Recovery Alliance (CRA), which founded the Windy City’s first needle exchange. CRA was also the first organization in the world to widely distribute the overdose reversal drug naloxone—and train drug users to save each other’s lives by using it. Naloxone (also known as Narcan) is a pure antidote to opioids: it restores the drive to breathe in overdose victims but must be given rapidly to be effective. (If used in error, it is safe: it won’t hurt people with other medical problems and typically works even if opioids have been combined with other drugs.) CRA’s approach is called harm reduction, and it defines recovery as making “any positive change.” This means that anything from starting to use clean needles to becoming completely abstinent counts. From this perspective, if someone quits smoking crack, gets a job and reconciles with her family, she counts as being in recovery—even if she stills smokes marijuana daily. Or, if someone goes from drinking a bottle of Scotch a day to having a daily glass of wine—or from drinking daily to binging only on weekends—these too are positive changes, not just “active addiction.” Here, recovery is a process, not an event. It’s difficult to learn any new skill without trial and error, and this includes developing coping skills to manage or end drug use. For most people, even with behaviors short of addiction, big changes take time. This broad definition obviously includes people who take addiction medications. Doing so is a positive change because it dramatically reduces the risk of death, even for those who continue to take other substances. Moreover, those who do quit nonmedical use and stabilize on an appropriate dose of these meds can drive, connect with others and work as well as anyone else. They are not intoxicated or numb—just like people taking other psychiatric drugs as prescribed. While patients on methadone or buprenorphine remain physically dependent on medication to avoid withdrawal, they no longer meet criteria for addiction. According to psychiatry’s diagnostic manual and the National Institute on Drug Abuse, addiction must include compulsive behavior despite negative outcomes—it’s not simply needing something to function. Of course, for people steeped in traditional abstinence-oriented recovery, CRA’s “any positive change” definition can be challenging. In the 12-step world, members who have maintained continuous abstinence for many years are revered—the longer their time away from alcohol and other drugs, the higher their status tends to be. The lure of such social acclaim helps some avoid relapse. Granting the status of “recovering” to those who have not quit entirely seems unfair from this perspective. However, it could save lives. In 12-step programs, people who break continuous abstinence—even for just one day after 20 years—are seen as returning to square one, and their “sober time” and its associated status is completely erased. Research shows that having such a binary view of recovery can actually make relapses more dangerous. That’s because people figure that, since they’ve blown it already, their small slip might as well be a massive spree. Since most people do relapse at least once, moving away from the idea that only continuous abstinence matters—not quality of life, not the ability to maintain relationships and contribute to society—would likely be healthier for everyone. But there’s another way to reconcile conflicting views of recovery, which preserves traditional ideas for those who prefer them. That is, simply define it for yourself and let others do likewise. If you’ve heard someone identify themselves by saying, “I’m a person in long-term recovery and for me, that means abstinence,” you’ve seen this idea in practice. My own perspective has changed over time. From 1988 through 2001, I was continuously abstinent from drugs other than caffeine, including alcohol. Since then, I have used alcohol and cannabis responsibly, without difficulty. However, I have no illusions that I could moderate either cocaine or heroin use—so I continue to avoid these drugs and count myself among the recovering. Now, though, I suspect that my recovery probably started before my abstinence—when I was taught to use bleach to clean my needles in 1986 and began to fight to get HIV prevention information and equipment to other injectors. That positive change likely helped prepare me for further transformation, including seeking rehab. It almost certainly helped me avoid AIDS. What really matters is not whether someone recovers via medication or moderation or 12-step programs or anything else. It’s that, like me, most people do get better. And even more of us can if we recognize and support many roads to recovery. from https://ift.tt/3F0fDQJ Check out https://takiaisfobia.blogspot.com/ One Monday in late February 2020, Lauren Gardner was working frantically. The website she’d been managing around the clock for the last month—which tracked cases of an emerging respiratory disease called COVID-19, and presented the spread in maps and charts—was, all of a sudden, getting inundated with visitors and kept crashing. As Gardner, an associate professor of engineering at Johns Hopkins University (JHU), struggled to get the site online again, an official in the Trump Administration falsely claimed on Twitter that JHU had deliberately censored the information. “Seems like bad timing to stop helping the world with this (previously) useful resource,” read the tweet from Ken Cuccinelli, then the Acting Deputy Secretary of Homeland Security. “Here’s hoping it goes back up soon.” It was a hypocritical allegation coming from an administration official. At the time, President Donald Trump was downplaying the virus’s potential to spread, and government agencies weren’t making public any of their own comprehensive data to support that position. What’s more, federal agencies were looking to JHU to find out what was going on. When former Vice President Mike Pence toured the government’s COVID-19 operations center the same week of the accusatory tweet, a huge projection of Gardner’s website illuminated one wall. The worldwide case count, glowing bright red, stood at 82,548. The total death count was 2,810. (As of September 28, there have been more than 230 million reported cases worldwide and over 4.7 million reported deaths.) A hotter head, especially one as sleep deprived as Gardner, may have gone off on a social media tirade against the administration. “I’ll just say, it was annoying,” she says now, reflecting on the day. The truth is that she was too busy—and too good natured—to let the comment get under her skin. 
Early in the pandemic, JHU’s COVID-19 dashboard filled an enormous void. It was the first website to aggregate and publish case and death figures in near real time: whenever a municipality updated its tally, the numbers would show up on the dashboard. And, critically, the data were easy to interpret and fully accessible to anyone. Even though the statistics were incomplete, they were the most meaningful measurements of the pandemic at the time, and enabled health experts and policymakers to cite the data when writing guidelines impacting how people work, travel and interact. The site helped media outlets, including TIME, report on the latest COVID-19 hotspots and trends. And it meant citizens could understand the risk of spread in their states and counties. “People were so desperate for information,” recalls Gardner. “We put it out there in a way that was just so easy to interpret.” It was a trailblazing endeavor. After JHU dashboard’s launch, U.S. agencies like the Centers for Disease Control and Prevention (CDC) and the Department of Health and Human Services (HHS) eventually launched their own robust COVID-19 data websites simple enough for any teenager, grandparent or armchair statistician to dive into. Other countries, along with U.S. states and cities, as well as other organizations and universities followed suit. As these dashboards proliferated, they reset the public’s expectations for what health data should look like. Today, the people trust, and even take for granted, that timely, detailed and nicely packaged information is available at the tap of a screen. Disappearing dataBut it’s a fragile system. Recently, some U.S. coronavirus data have become harder to find or completely unavailable. That’s not because the websites are crashing from traffic overload, but because some U.S. states are consciously removing information or shutting down their dashboards entirely. Gardner and other members of the JHU team are dismayed by the reversal. 
“The fact that they created it, the infrastructure sits there, they put resources towards it, and then they decided not to make it sustainable is really concerning and just disheartening,” says Gardner. “It’s like they started going down the right path, and then pulled back.” By the first week of June, 24 U.S. states were reporting cases and deaths less than seven times a week and four states were reporting only one to three times a week, according to JHU. More states followed suit, even as cases began to rise again due to the Delta variant. Currently, 36 states have pulled back from daily reporting, and seven of them are reporting only three times a week or less, including hard-hit Florida, which is reporting weekly. https://datawrapper.dwcdn.net/YC8Si/2/ And that’s just for cases and deaths. Other, more detailed metrics are evaporating. Georgia stopped providing data on prisons and long-term care facilities. Nebraska eliminated county-level data, citing a strict privacy statute intended to protect citizens from being identified. (An executive order from the governor had lifted those restrictions during the state of emergency, which ended in June.) Florida dropped its granular reports on pediatric hospitalizations, prompting a state representative and media outlets to sue the health department for them to be reinstated. Not all of the metrics have disappeared completely. Some are still being reported at the federal level. But the public must weed through nationwide data to find their community—if they know where to look. Want to see death trends among seniors? Dig around the CDC site. Hospital capacity? Best bet is HHS. Cases by nursing home facility? Head over to Centers for Medicare and Medicaid Services. Plus, federal agencies are often the last stop on the information pipeline, leading to reporting delays and discrepancies. For instance, the CDC cannot officially report a COVID-19 death until the death certificate has been submitted and processed with the agency—a process that can take weeks—which means that the agency’s validated COVID-19 mortality stats don’t align with the states’ stats. The upshot is that the COVID-19 picture is getting fuzzier, even as the highly contagious Delta variant rages across the country, straining health care systems and killing thousands every day. “We should be doubling down on this infrastructure instead of this how-can-we-quickly-get-the-genie-back-in-the-bottle kind of thinking that we’re seeing in some places,” says Beth Blauer, the associate vice provost for public sector innovation at JHU, who has worked alongside Gardner to refine, analyze and contextualize the dashboard data. Gardner is similarly frustrated. “If you don’t know what’s going on, then the virus gets out of control and then it’s harder to get back in control.” Taking on an information voidAggregating public health data is no picnic. The world, and the U.S., rely on a hodgepodge of (often outdated) surveillance systems. Like a game of telephone, there’s a risk of error and delay each time data are relayed between health providers, labs, and various health departments and agencies. Compounding the problem is the longstanding public-health worker shortage. “The lack of standards and the lack of technology and information systems that are interoperable and can exchange standardized data—that has really been one of the barriers to having timely, complete information in this pandemic,” says Paula Yoon, director for health informatics and surveillance at the CDC. Gardner, who studies the systems science of infectious diseases—that is, how they spread based on the nature of the pathogen in question, human behavior, demographics and other factors—is not an epidemiologist or a public health official. So like most Americans, she didn’t fully realize the extent of the health system’s problems until she was thrust into a position where she had to grapple with them. Read More: 73 Experts Offer a Blueprint for Preventing Another Pandemic On the morning of Jan. 21, 2020, Gardner’s graduate student Ensheng Dong mentioned that he had been tracking the coronavirus in real time. They came up with an idea to plot the roughly 500 cases recorded so far on a map. Dong, a native of China, had lived through SARS, and so was concerned about his family and friends back home. Gardner knew it was a unique opportunity to model, or predict, the spread of a new disease. Because models are only as good as the data fed into them, a real-time, open-source case counter might give them—and other researchers—more insights than the World Health Organization’s once-daily summary reports that often showed lagging data. By the next morning, the dashboard was live. It featured a map of Asia, with land masses in black, punctuated by bright red dots, some as small as pinholes. China had one in nearly every province—the largest dot was in Hubei, at that point the epicenter of the outbreak. From that day on, the red dots would multiply, jump across oceans and balloon in size until whole continents turned from charcoal black to firetruck red. 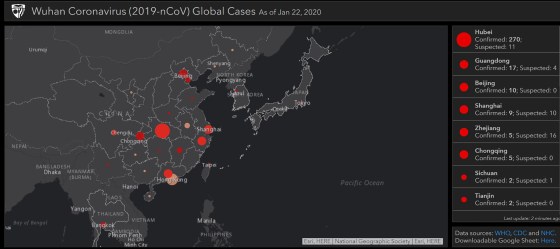
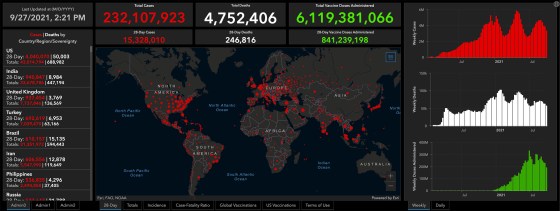
Of course, Gardner wasn’t anticipating any of that, and she didn’t expect the public to be so captivated. “I was like, oh, maybe there will be a few dozen people that are interested in using this,” she says. Instead, the dashboard went viral, growing to one million unique daily visitors by mid-February and 3.5 million in April. From the beginning, there was pressure to keep the map current and accurate. At first, Dong manually collected and inputted the data from Chinese government sites and dxy.cn, an online community for health care professionals. For other countries, Dong relied on media reports based on statements from local public health authorities, because it would often take days for those numbers to show up in the official state or country tally. He scoured social media for news of cases and even crowdsourced the job—people around the world would email him, flagging fresh numbers from their hometowns. Gardner managed the thousands of messages and requests from journalists, health professionals and governments. She also oversaw technical aspects of the project, making sure the interactive map and the back-end database functioned properly. Meanwhile, she began assembling a small team of volunteer grad students to work with Dong, who had dropped a class to keep up. Gardner shelved her other research projects. Neither of them got more than a few hours of sleep each day—the global nature of the project meant that the data and inquiries needed attention at all hours. “I clearly remember having a dream that I put some British overseas territory under the French [count],” says Dong. “I instantly woke up and jumped to my computer and checked if the territory was in the right category. I was so nervous during that period.” Then, the red dots began to spread across the U.S. JHU went into lockdown with the rest of the country. Gardner was running on adrenaline. The information JHU was providing filled a massive void—no other national health ministry was updating case and death counts as frequently. But the dashboard’s popularity threw Gardner into a position of authority that she hadn’t signed up for. “I wouldn’t have proactively sought out this job,” she says. “I am a data user, not a data provider.” 
The team had to smooth out imperfections on the fly. Take, for instance, the single red dot in the middle of the Atlantic that represented cases without geographic data. It lived at 0 degrees both latitude and longitude, a point known as “Null Island” by geocoders because it represents data that is empty, or null. It wrongly gave the impression that there was a growing outbreak some 300 miles south of Ghana. Another time, when virus-positive passengers on a cruise ship were repatriated to the U.S., the team generically assigned them to the “United States.” That put a dot in the middle of the country, prompting residents of a Kansas town to demand JHU offer an explanation and a correction. Still, fixing the map’s presentation was easy compared with fixing the data collection problems. As the virus spread, manually updating the dashboard became unsustainable: by the time the team had circled the world, the data would be outdated and they’d have to start again. So Gardner assembled a willing and dedicated geek squad. One group of coders at JHU’s Applied Physics Laboratory offered to write computer programs that could automatically scrape data from sources, organize it into a clean format, and flag any anomalies for a second, manual look. ESRI, the company providing the dashboard’s map software, upgraded the website incrementally over several months, working to stabilize it without having to ever take it down entirely. “It was the equivalent of trying to change the tires and the engine of a moving car, and then repainting it and then adding a trailer to it,” says Sean Breyer, an ESRI program manager who was integral in the JHU effort. “There was no way to stop.” Read More: Inside the Group Peddling a Bogus COVID-19 Treatment Also joining the effort was Lainie Rutkow, a senior advisor in JHU’s president’s office, who corralled experts from around the campus to lend knowledge and insights. One was Blauer, who took on data analytics, built charts and wrote about trends. Another was Jennifer Nuzzo, a senior scholar and associate professor of epidemiology at the Bloomberg School of Public Health, who spearheaded an effort to publish testing numbers and positivity rates. The cogs behind what became known as the JHU Coronavirus Resource Center began to turn more smoothly, but one systemic problem remained: The data were riddled with disparities and anomalies. Health departments in the U.S. and abroad were stretched beyond capacity and couldn’t stop to establish uniform protocols for what counted as a case or a death. In the U.S., some jurisdictions included positive antibody tests in their case counts. Others did not. Some tallied probable deaths that hadn’t been verified. And many would do data dumps as they changed methodologies or cleared a backlog of health records. For example, one day in mid-April, 2020, one of JHU’s sources added three digits to Okaloosa County, Florida, boosting its case count a thousandfold overnight. Another: this past spring, the United Kingdom reported negative case numbers, after finding that thousands of people had received a false positive test. And just a month ago, France made several back-dated revisions to its nursing home death count, which hadn’t been updated for six months. https://datawrapper.dwcdn.net/hFSHz/1/ Still, the team never took liberties: they reported only what the health agencies and other reliable sources published and worked as quickly as possible to deal with irregularities. A messy and testy data revolutionJHU’s real-time counters have never shown a complete picture of the actual case and death counts. For one thing, official reported cases might not show the true extent of viral spread: many people who have likely contracted COVID-19 have probably not been tested, particularly if they were sick in the early days of the pandemic or if they never exhibited symptoms. Moreover, the emails, faxes and pdf documents sent from labs to the health departments must be manually processed, causing delays, especially when they are incomplete. Unlike JHU, which reported information as soon as it became available, the CDC waits on the states to send information, which can take weeks or months. Public health experts say there was little chance that the CDC was prepared to monitor the virus from the outset. “Our public health data are way outdated,” says Marcus Plescia, the chief medical officer at the Association for State and Territorial Health Officials. “We need to modernize the infrastructure so that our systems work and communicate well across states and compile national data more quickly. JHU did that when CDC wasn’t able to. JHU had the ability to be facile and nimble and work with what was there in a quick time frame.” Yoon, the CDC surveillance director, agrees that systems are overdue for an overhaul. “The lack of investment in the public health infrastructure for these past 10, 15, 20 years have left us unable to have the timely and complete data that we—CDC and state and local entities—need to respond more quickly,” she says. 
In an effort to catch up, the CDC launched, in April 2020, an online COVID-19 Data Tracker, which eventually expanded into a comprehensive website chock full of charts and the latest research. But getting data onto the site was a long and bumpy process. It took until May 2020 for the agency to start reporting in the same fashion as JHU—scraping case and death data directly from state websites, rather than waiting for the health departments to send them. For county data, the agency relied on the nonprofit USAFacts.org until September of that year, when it was able to scrape its own county-level data. The delay wasn’t just bad for the CDC’s reputation; the absence of a central government data hub left the impression that JHU was the gold standard. Health agencies that were struggling to process the tsunami of health records felt pressure to conform to JHU’s priorities because the dashboard was so popular, says Jeff Engel, formerly a executive director at the Council of State and Territorial Epidemiologists (CSTE) and a North Carolina state health official, who recently retired. Take, for example, JHU’s tally of “recovered cases.” “Once Hopkins started reporting it, our states had to come up with the metric and were making things up because this was something that Hopkins made up,” says Engel. But it was a tally so woefully incomplete and ill defined, as to be meaningless. “Our staff were going, we don’t even understand what this number means,” says Engel. “And also, why would you even need it?” Gardner says the numbers were sourced from a few places around the world that were reporting them. She admits that it was a shaky data point, and the team eventually dropped it from the dashboard. Read More: Why COVID-19 Might Be Here to Stay—And How We’ll Learn to Live With It But bad data have real-world consequences. For instance, U.S. states restricted travel from other states based on the positivity rate—but positivity rate is a metric derived from testing, and testing volumes and the types of tests being calculated varied from state to state. The JHU team was well aware of the problem. “We were having calls with state departments of health and governor’s offices where they were like, ‘why do we look so horrible?’” recalls Blauer. “We were just so frustrated with the data landscape and the fact that there were no standards.” The fight to stay aheadTo establish effective public health policies, surveillance data must be timely and accurate, so trends become apparent as they’re happening—not after the fact. That may seem obvious, but that effort takes a lot of time, resources, coordination—and a bit of intuition and a leap of faith that the data-collection efforts will pay off. Such efforts do often pay off. Consider that metrics like age and race of those infected or killed didn’t exist early in the pandemic. Once those data became available, it became obvious that certain populations, like elderly and minority communities, were getting disproportionately hit. These essential findings lead some states to deploy health education media campaigns and testing centers in the most vulnerable neighborhoods, but those initiatives came too late for many. We didn’t learn our lesson from that experience. When vaccines started rolling out, demographic data-collection was spotty at best. As states made a greater effort to report it, it became clear that many communities of color were getting left behind. https://datawrapper.dwcdn.net/MR0fd/1/ Spurred on by those data, many jurisdictions then launched vaccination outreach programs focusing on those communities. Cities deployed mobile vaccination vehicles to the most underserved neighborhoods, for example, and also collaborated with Black churches and charitable groups to serve those without transportation or internet access. Nevertheless, there remain areas in which lack of data continues to prolong and even exacerbate the pandemic in the U.S. For example, in the spring, virology and immunology experts warned that slow and spotty data around emerging virus variants would hamstring our ability to stay ahead of the disease as it mutated. Sure enough, as the variant was taking hold of the southern U.S., very few positive test swabs were being genetically sequenced to detect which variants were circulating, leaving a huge blind spot. “We were caught with our pants down because by the time we ramped it up, it was too late,” says Engel. As more and more places stop reporting vital information, history is bound to repeat itself. Sharon Dolovich, who runs a UCLA-based COVID-19 monitoring project for inmates, feels her efforts are increasingly thwarted by corrections departments that have stopped reporting. Despite evidence that prison populations have been especially susceptible to outbreaks, states claim that tracking COVID-19 is not necessary because the situation is under control. That rationale is “a self-fulfilling prophecy,” says Dolovich. “The story you tell becomes reinforced, and then it provides a justification for not doing anything.” 
It’s not all bad news, though. In many respects, the COVID-19 data revolution has spurred major infrastructural leaps that health departments can build upon going forward. For example, some of the slow and leaky information pipelines connecting health care providers and state health agencies have already been upgraded. Nearly 10,000 providers now have an electronic case-reporting system, which automatically alerts agencies to cases of COVID-19 as soon as they are diagnosed, along with other information on the patient’s health record, like age and race. That’s a huge jump from the 187 providers that had such a system at the start of the pandemic. What’s better, the system works for any other reportable conditions like HIV, measles and rabies and can be adapted for future diseases. The U.S. National Institute of Health, meanwhile, launched a data portal in the summer of 2020 to collect and standardize electronic health records from COVID-19 patients. In a world of many different health record formats, the project has been an enormous undertaking. “We took a card from JHU’s playbook,” says Joni L. Rutter, acting director of the National Center for Advancing Translational Sciences at NIH, which oversees the project. “It’s not sexy work, but data need to have integrity.” Researchers are using the data to find which patients are most at risk for severe disease, and which treatments work best for each patient based on their health profile. Their findings are publicly available. And though the project is only authorized for COVID-19 research, Rutter sees huge potential beyond the pandemic. “Now that this shows proof of principle, electronic health record research might be here to stay and I think it should be,” she says. “The question is, how can we apply it to build something bigger?” It’s difficult to reconcile such formidable progress and future potential with the ongoing struggle to track the ongoing health crisis. At JHU, Blauer recently launched the Pandemic Data Initiative, a digital hub sharing the university team’s insights and best practices, as well as its ongoing challenges. She confesses that the site serves as “a place where I air a lot of my frustrations with the data.” In a recent post, she highlighted all the problems and inconsistencies with how states are tracking breakthrough cases. (The CDC stopped tracking breakthrough cases in May.) 
Gardner, too, remains committed to fixing the ongoing problems and keeping information open and transparent. “The problem is, if we just keep staying behind [the virus], then you keep giving it chances to bring us back to step one,” she says. “The worst thing to do is to not know what’s going on.” from https://ift.tt/3ulyRvl Check out https://takiaisfobia.blogspot.com/ WASHINGTON — President Joe Biden will receive his COVID-19 booster shot on Monday, days after federal regulators recommended a third dose of the Pfizer vaccine for Americans age 65 or older and approved them for others with preexisting medical conditions and high-risk work environments. The White House said Biden, 78, would deliver remarks and receive the additional dose at 1 p.m. Monday. Biden got his first shot on Dec. 21 and his second dose three weeks later, on Jan. 11, along with his wife, Jill Biden. It was not immediately clear whether the first lady would also receive the booster dose on Monday. Speaking on Friday after the Centers for Disease Control and Prevention and the Food and Drug Administration authorized the Pfizer booster, Biden told reporters, “I’ll be getting my booster shot. It’s hard to acknowledge I’m over 65, but I’ll be getting my booster shot. ” Vice President Kamala Harris, 56, received the Moderna vaccine, for which federal regulators have not yet authorized boosters — but they are expected to in the coming weeks. from https://ift.tt/3ALygVX Check out https://takiaisfobia.blogspot.com/ In Aly Raisman: From Darkness to Light, the former Olympian confronts her most challenging task yet: recovering from the sexual abuse she experienced while training as an elite gymnast. Raisman is one of more than 200 survivors of sexual abuse by former USA Gymnastics’ team doctor Larry Nassar; in the documentary, which airs tonight on the Lifetime channel, she sits down with other survivors who were sexually abused by trusted members of the community as children, and breaks through the walls of fear, intimidation, ignorance and prejudice that keep such abuse in the dark. The journey she chronicles is both theirs and her own, and part of what makes the documentary so powerful is Raisman’s vulnerability and her transparency regarding her own struggles to process what she went through. In the special, Raisman says that she still finds hearing about other people’s experiences “triggering;” and in one scene, we see her quietly leaving the courtroom as another survivor details the traumatic experience of her abuse to the California state legislature. “It was important for me to do this because most survivors aren’t supported the way I was supported,” Raisman tells TIME. “I’ve really learned the importance and the power of community and how crucial that is for survivors. I hope this special gives people a better understanding of how common sexual abuse is, and I hope it’s a reminder that no matter what you’re going through, there is a light at the end of the tunnel, you are going to be okay and there is help out there. I hope it starts the conversation, whether it’s at the dinner table, or at work, or in youth sports groups, and inspires people to ask questions and learn more.” Throughout the three-hour special, Raisman solicits advice from other sexual abuse advocates, like #metoo movement founder Tarana Burke, who counsels the retired gymnast to make sure she looks after her own mental health as she embarks on the journey to speak with other survivors. The documentary comes at a time when Raisman and other former USA gymnasts are still pushing for USA Gymnastics (USAG) and the U.S. Olympic and Paralympic Committee to take responsibility for failing to protect young athletes even after a few of them and their parents and coaches reported Nassar’s abuse to USAG. Raisman, along with Olympic teammates McKayla Maroney and Simone Biles, and national team member Maggie Nichols, testified on Sept. 15 before the Senate Judiciary Committee about the FBI’s flawed investigation of the Nassar sexual abuse reports. Raisman says she felt it was important to testify, despite the pain of reliving her own abuse experiences. “I know it’s so much bigger than myself,” she says. It also took a lot to overcome her fear of letting others down. “There is so much pressure on us to deliver good testimony,” says Raisman. “If we don’t, there’s the pressure of, ‘Will this move the needle? Will people listen? Will people hear us? Will this effectuate change?’” To ensure that more young children don’t have the same experience that she had, Raisman is working with the non-profit Darkness to Light, which seeks to better equip adults to recognize child abuse, and to talk to children about it—even in cases where children are too young to recognize it themselves, or, if they do, too fearful to say anything. She is also intent on chronicling not just the trauma but also her experience healing. “The thing that has helped me the most in my healing is being patient with myself, and giving myself permission to have tougher days where I do feel really tired,” she says. “I used to be hard on myself, and I still am, by I’m trying to be kinder to myself.” The special first aired on Lifetime on Sept. 24. from https://ift.tt/2Zp83yI Check out https://takiaisfobia.blogspot.com/ You can say one thing for Professor Mark Haub: He knows how to make a lesson stick. Haub, who teaches nutrition at Kansas State University, wanted to prove to his students that weight loss is simply about calories. So, for 10 weeks, the professor proceeded to eat an 1,800-calorie diet consisting of a Twinkie every three hours. He also dined on Doritos, Little Debbies, sugary cereal and other junk food. When he started, Haub tipped the scales at 201 pounds, which for his height was considered overweight. By the end of his snack-food spree, he had lost 27 pounds, putting him at a svelte 174. The story went viral, with the media dubbing Haub’s eating plan the Twinkie Diet. Undoubtedly some who heard the news eagerly stocked up on the spongy yellow snacks. But Haub’s intention wasn’t to urge people to eat more Twinkies. The point, he said, was that he had consumed 800 fewer calories daily than the number needed to maintain his weight. In other words, the key to weight control is counting calories: If you take in fewer than you burn, you lose weight. It’s that simple. Haub’s message has been standard advice for more than a century. According to many experts, it all boils down to straightforward math: Calories in minus calories out. Countless millions who struggle with their weight heed this message, dutifully tracking their calorie intake. But eventually many discover that all the counting is in vain. One reason is that calorie counts aren’t always accurate. In fact, the Food and Drug Administration (FDA) allows numbers on nutrition labels to be off by as much as 20 percent, and usually the error is an undercount. That means, for example, that ice cream claiming to have 180 calories per serving may actually have 215. Making matters worse is the widespread problem of unrealistic serving sizes. With ice cream, if you eat a cup (a normal amount) rather than 2/3 cup (the usual serving size), you could be getting as many as 325 calories instead of the 180 listed on the label. Listed calories may also be wrong because of the way our bodies digest certain foods. Take almonds, for instance. Nutrition labels show them to have up to 170 calories per ounce. But this number doesn’t take into account the fact that almonds pass through the intestines partly undigested. As a result, the body doesn’t absorb all 170 calories. The actual count, according to research, is 129—a sizable difference. Accurate or not, calorie counts aren’t available for everything we eat, so we sometimes have to rely on our own estimates. And according to research, these numbers are notoriously unreliable. For example, in a survey of 2,200 adults, consumers’ guesses about calories in popular restaurant foods ranging from pancakes to onion rings undershot the reality by an average of 165 calories. Unconscious biases can further skew our calorie estimates. For instance, there’s the “health halo” bias, which makes us more likely to underestimate calories in foods that are marketed as healthful. Online calculators, meanwhile, can tell you how many calories you expend each day, but it’s at best an approximation. Wearable devices are also an option, but research shows that their results are unreliable. Arriving at an accurate number is difficult because the calculation is complex, involving how much energy we need for basic functions like breathing and circulation at rest (known as basal metabolic rate, or BMR); how much we burn during everyday activities and exercise; and how much through digesting food (the thermic effect of food). A host of other factors, including age, gender, weight and body fat, play a role. Given all the challenges of accurately calculating how many calories we need and how many we consume, it’s unreasonable to expect counting calories to be effective as a weight-loss strategy. Read more: 9 Common Myths About Exercise The difficulty is reason enough to shun calorie counting. But there’s also an even bigger problem: Tallying calories fails to take into account other variables that can affect how much we weigh. As we reduce calories and lose weight, biological changes kick in to preserve body fat and protect us from starvation. One such adaptation is a change in metabolism. The body of a lighter person has a lower BMR than that of a heavier person. As we shed pounds, we burn even fewer calories than expected for a person of our reduced size—a phenomenon that scientists call adaptive thermogenesis. In essence, our bodies become more fuel efficient, making it increasingly difficult to shed more pounds and to maintain weight loss with the same number of calories. Unfortunately, this evolutionary gift, designed to keep us alive in times of scarcity, isn’t something we can switch off or send back when we don’t need it. Our genetic makeup also affects weight regulation. As evidence, look no further than those maddening people who seemingly can eat whatever they want and never gain an ounce. Conventional wisdom has it that such individuals are blessed with “good genes,” and research involving twins shows genes do affect how our bodies respond to calories. In one study, for example, researchers observed 12 pairs of male identical twins for four months, supervising their every move. (Yes, the twins agreed to this!) The subjects were fed 1,000 calories a day more than their normal intake, and physical activity was limited. As you would expect, they gained weight. But the amount varied, ranging from about 10 to 30 pounds. What’s more, the difference in the amount of weight gained was much smaller between twins in a pair than among different twin pairs. In other words, twins in each pair experienced relatively similar increases in weight, suggesting that genetic factors influence how easily we put on pounds. Similar research suggests genetics affects how easily we lose weight as well. Yet another possible contributor to weight is the mix of microbes in our gut. This community of bacteria, viruses and other microorganisms, known as the microbiota, helps break down food and extract energy from it. Studies show that the microbiota of obese people differs from that of lean individuals. Though this research is still in its infancy, it suggests that two people can eat the same amount of the same food and experience different effects on their weight depending on the makeup of their microbiota. Those whose gut microbes harvest more energy from food may be more likely to pack on pounds because it’s the calories we absorb—as opposed to the ones we ingest—that matter when it comes to our weight. Read more: Fast Food Calorie Content Has Steadily Increased Over the Past 30 Years Counting calories can be effective for weight loss in the short term, and it may work long term for some. But for the vast majority of people, it eventually not only fails but also can do harm. For starters, it can detract from the pleasure of eating, turning meals into a tedious exercise of tallying and food weighing. This routine can be stressful and may contribute to an unhealthy relationship with food that makes it even harder to achieve and maintain a healthy weight. What’s more, calorie obsession can lead to food choices and eating habits that undermine your health. Not all calories are the same—50 calories of broccoli doesn’t equal 50 calories of jelly beans—and a low-calorie diet is not necessarily a healthy one. Focusing only on calories can result in too little of things your body needs and too much of things it doesn’t need. So what’s the alternative? While it’s good to keep a general eye on calories, don’t fixate on them. Instead, pay attention to the overall quality of your diet, emphasizing vegetables, fruits, whole grains, beans, nuts, seafood and lean meats, while minimizing highly processed foods such as chips, cookies, fried foods and sugary beverages. To say our bodies’ weight-regulation mechanisms are complex is an understatement. After many decades of research, there’s still much that scientists don’t understand. So it defies logic that a simple food-scoring system conceived in the 19th century should be adequate for capturing this complexity. Yet calorie counting and calorie math continue to be mainstays of weight-loss efforts. It is not surprising that our society’s preoccupation with this inadequate and error-prone metric has yielded such poor results. What is surprising is that we nevertheless continue to give it so much weight. Adapted from Supersized Lies: How Myths About Weight Loss Are Keeping Us Fat – and the Truth About What Really Works by Robert J. Davis. Copyright © 2021 by Robert J. Davis. Reprinted by permission of Everwell Books. from https://ift.tt/3AIoKTz Check out https://takiaisfobia.blogspot.com/ CDC Panel Outlines Who Should Get Pfizer-BioNTech COVID-19 Booster Shots And Who Should Wait9/23/2021 In a four-part vote, a Centers for Disease Control (CDC) committee today recommended booster shots of Pfizer-BioNTech’s COVID-19 vaccine for people 65 years or older, as well as anyone over 50 with certain underlying medical conditions. The committee also recommended allowing people 18- to 49-years-old with underlying medical conditions to receive a booster if they desired, based on individualized decisions about the benefits and risks to them. But in one of its four votes, the committee decided against recommending booster shots for adults working in high-risk settings such as health care and school systems. That vote went against the Food and Drug Administration’s (FDA) decision on Sept. 22 to authorize a booster dose of the Pfizer-BioNTech vaccine for people over 65-years-old, those ages 18 to 64 who are at higher risk of getting the disease, such as people with health conditions that weaken their immune systems, as well as health-care, front-line and emergency workers and teachers among others whose jobs make them more vulnerable to getting exposed to COVID-19. The FDA recommended that the booster be given at least six months after an individual receives the second of the two-dose vaccine to all of those groups. It was up to the CDC committee that met today, however, to determine how that authorization should be translated to the eligible groups. While the CDC’s Advisory Committee on Immunization Practices (ACIP) often follows the recommendation of the FDA, it doesn’t have to—the CDC’s mandate, which focuses more on public health goals, differs slightly from that of the FDA’s, which is more centered around evaluating safety and efficacy. “What we need to be doing is to look at the totality of evidence and think about what makes the most sense using the tools we have to protect as many people as possible, and how to operationalize this considering other things like feasibility,” Dr. Beth Bell, professor of global health at the University of Washington, reminded the committee. While the members generally agreed about recommending a booster for those over 65, they were divided about the necessity of the booster at this time for younger people, even those at higher risk of COVID-19 exposure because of their jobs or other circumstances. So far in the U.S., 220 million doses of Pfizer-BioNTech have been administered, and 27 million people over age 65 years have received both doses of the vaccine. They were among the first to get vaccinated after Pfizer-BioNTech’s vaccine was approved, and now show the first signs of waning immunity—which is why the committee prioritized them to receive a booster dose to strengthen that protection. In discussing whether boosters should also be extended to people with underlying medical conditions and those whose jobs might put them at higher risk of exposure, some members noted that authorizing the booster for these populations would raise concerns about health care equity, arguing that it could deepen gaps in coverage as those without access to care who remain unvaccinated continue to miss out on the opportunity to get protected against COVID-19. Ultimately, the committee decided to fully recommend the booster for everyone over 50 with underlying medical conditions, but to offer a less full-throated endorsement for those aged 18 to 49 with such conditions. For them, the ACIP is recommending that people choose for themselves whether they want to get a booster dose. The CDC committee then deviated from the FDA when it came to people like health care workers under age 65 whose jobs might put them at increased risk of exposure and infection, noting that there isn’t strong evidence to suggest that they, if they are vaccinated, are experiencing higher rates of severe disease. The data show that the currently approved two doses of Pfizer-BioNTech continue to provide good protection against COVID-19, especially against severe illness, hospitalization and death. In particular, while Pfizer-BioNTech did provide the FDA and CDC with some data showing that a booster shot raised antibody levels among young people, those data only followed those people for a few months. And committee members also raised the point that, while apparently rare, some young males can develop a rare inflammation of the heart tissue as a side effect of getting the vaccine. That’s why some of the members emphasized that their support of the booster today was an interim one, for an initial group of people, based on the available data available. “This is the beginning of a lot of activity around booster doses,” said Bell. “At this moment, given the lack of evidence for marginal benefit of boosters in people in certain groups who have received the Pfizer-BioNTech primary series—it’s too narrow, and too soon, and given the potential risks or adverse outcomes it’s worth waiting until we know a little more about what we are doing, and we can better assess the opportunity costs, and the unintended consequences.” The CDC committee was instructed that its booster decision would only apply to those who had been inoculated with the Pfizer-BioNTech vaccine to begin with, since the FDA only reviewed data on that population. Some committee members, however, argued that the decision should also apply to people who initially got Moderna or Johnson&Johnson-Janssen shots in order to be fair and equitable. (Moderna has also submitted a request to the FDA for authorization of a booster dose of its vaccine, but the agency has not yet reviewed those results.) Such mixing and matching of doses, however, isn’t supported by currently available data. Some committee members, like Dr. Nirav Shah, director of the Maine Center for Disease Control and Prevention, argued that allowing mixing and matching of boosters would help more people to get vaccinated. He noted the burden of asking people to keep track of their vaccination records, and of asking those administering vaccines to validate those records. Dr. Sarah Long, professor of pediatrics at Drexel University College of Medicine, added that limiting the booster to only those who initially got the Pfizer shot would be especially unfair to those living in long-term-care facilities who received an initial series of Moderna vaccine and would not be able to get a booster while their neighbors who received the Pfizer shot would be vaccinated again. “I don’t understand how later this afternoon we will say to people over 65 years that you are at risk of severe disease and death, but only half of you can protect yourselves right now,” she said. But, as Dr. Doran Fink, from the FDA’s office of vaccines research and review told the committee, “there is currently no data available to inform interchangeability of authorized COVID vaccines, either for completing of the [two-dose] primary series or for use as a booster dose.” Because of this lack of data, the CDC panel ultimately decided that only people who previously received two doses of the Pfizer-BioNTech vaccine should receive a booster dose of the Pfizer-BioNTech shot. This, however, is another area where today’s recommendation may not be the final word: study results on such mix-and-match approaches are expected imminently; researchers said the trial was completed, and the data have been submitted to a journal for publication. The committee stressed that, for most people who have been vaccinated, the current two-dose regimen still provides strong protection against SARS-CoV-2, including against the Delta variant. “Our decision today is not about who deserves a booster but who needs a booster,” said Dr. Matthew Daley, senior investigator at Kaiser Permanente Colorado. “If you are in a group for whom a booster isn’t recommended today, the reason the booster isn’t recommended for you today is because the vaccine efficacy against serious outcomes is already high for those who have gotten the primary series.” from https://ift.tt/3ACiFrR Check out https://takiaisfobia.blogspot.com/ When Joe Silverman developed Crohn’s disease at age 21, the symptoms started out mild. While the sight of blood in his stools initially freaked him out, what really bothered him was the frequent abdominal pain and bloating that occurred as his condition progressed to moderate and then severe. Dietary changes didn’t make a difference, so he began taking prescription oral anti-inflammatory drugs that are often used to treat certain bowel diseases, which alleviated but didn’t eliminate his discomfort. He started using prescription steroid suppositories to cope with flare-ups of the inflammatory bowel disease. Even so, “I didn’t feel well—my mind was cloudy and I was in pain,” says Silverman, now 47, the co-founder of the PSMC5 Foundation, which is dedicated to beating rare genetic disorders like the PSMC5 gene mutation (which his son has). So in 2013, he tried a new approach: he began getting intravenous infusions of an immunosuppressive drug at four- to eight-week intervals to reduce inflammation in the lining of his intestines. “It helped, but I still had nausea, brain fog, discomfort and trouble sleeping,” says Silverman. In 2018, he decided to try something different as an adjunctive treatment, with his gastroenterologist’s blessing: medical marijuana in the form of cannabidiol (CBD) and tetrahydrocannabinol (THC) capsules that he was able to purchase after getting a New York City medical-marijuana license. “Within an hour and a half of taking them, I felt better,” Silverman says. “The bloating and pain went down, and my appetite came back.” For centuries, marijuana, which is derived from the plant Cannabis sativa, has been used for both medicinal and recreational purposes. On the medicinal front, cannabinoids—a group of compounds that constitute the active ingredients in the marijuana plant—have been found to help alleviate chronic pain, as well as the nausea and vomiting that stem from chemotherapy for cancer. The U.S. Food and Drug Administration (FDA) has even approved specific cannabinoid products for chemotherapy-induced nausea and vomiting in cancer patients and to stimulate appetite in patients with AIDS who have lost weight. In recent years, there has been growing interest in the use of medical marijuana for gastrointestinal disorders, such as inflammatory bowel diseases (IBD) like Crohn’s and ulcerative colitis (UC). In a study in the December 2013 issue of the journal Inflammatory Bowel Diseases, researchers surveyed 292 patients with IBD at a major medical center in Boston about their use of marijuana and found that 12% were active users and 39% were past users. Among current and former users who used marijuana products for their symptoms, the majority felt that it was “very helpful” in relieving their abdominal pain, nausea and diarrhea. More recently, a 2018 study in the Journal of Pediatrics found that among 99 teen and young-adult patients with IBD, nearly one-third had used marijuana—and 57% of the users endorsed its use for at least one medical reason, most commonly relief of physical pain. “A lot of people perceive this as a more natural therapy and preferentially want this over immunosuppressants for inflammatory bowel disease,” says Dr. Byron Vaughn, an associate professor of medicine and co-director of the IBD program at the University of Minnesota in Minneapolis. But if anything, experts see the primary role for cannabis as an adjunctive therapy, not as a replacement for medications that are used to treat IBD and other GI disorders. Help or hype? Research investigating the effects of medical marijuana on various gastrointestinal disorders is limited, so there are many unanswered questions. Right now, one of the obstacles to this is the classification of cannabis as a Schedule I drug (along with heroin, LSD and ecstasy) by the federal government. This reality has inhibited research in the U.S. to evaluate the effects of cannabis on various gastrointestinal disorders as well as other medical conditions. And while the mechanisms of action aren’t completely understood, this much is clear: the human body has an endogenous cannabinoid system—one that originates inside the body—that comprises cannabinoid receptors, endogenous cannabinoids (lipids that engage cannabinoid receptors), and enzymes that are involved in the synthesis and degradation of the endocannabinoids. In particular, CB1 receptors are abundant in the central nervous system, while CB2 receptors are more prevalent throughout the gastrointestinal tract, explains Dr. Jami Kinnucan, an assistant professor of medicine in the division of gastroenterology and hepatology at the University of Michigan in Ann Arbor. A little background about cannabis: while it contains hundreds of compounds, the most well-known are THC and CBD. THC is responsible for marijuana’s psychoactive effects (that “high” sensation), whereas CBD is not psychoactive but seems to modulate the effects of THC, explains Dr. Christopher N. Andrews, a clinical professor of gastroenterology at the University of Calgary. As far as inflammatory gastrointestinal disorders go, the greatest symptom benefit seems to come from preparations that have a combination of THC and CBD, Kinnucan says. This is partly because while CB1 receptors are activated by THC, CBD and THC have a synergistic effect on CB2 receptors. “In patients with inflammatory bowel disease, studies have shown that the combination improves abdominal pain and decreases bowel movement frequency,” she says. What’s more, cannabis use appears to decrease emptying of the stomach and gastric-acid production, as well as reduce the movement of food throughout the gastrointestinal tract, notes Dr. David Poppers, a clinical professor of medicine in the division of gastroenterology and director of GI Quality and Strategic Initiatives at NYU Langone. As a result, cannabis use may improve the diarrhea-predominant form of irritable bowel syndrome (IBS), he says. Whether cannabis actually improves the underlying causes of GI disorders is less clear. “In the test tube, all cannabinoids have some anti-inflammatory effects,” says Dr. Jordan Tishler, an instructor of medicine at Harvard Medical School and president of the Association of Cannabinoid Specialists, a professional organization dedicated to education about cannabinoid medicine. “In human studies, if you look for blood markers of inflammation, you don’t see any change after using cannabis.” When it comes to treating IBD, “there isn’t a lot of evidence that cannabis really modifies the underlying disease process,” Tishler says. “But it treats the symptoms people have.” Other experts agree. “When you tease it out, this is more of a symptom-based therapy,” Vaughn says. “With IBD, there seems to be a calming effect on symptoms such as nausea, vomiting, pain and diarrhea.” Vaughn reports that he sees patients with Crohn’s disease get more of an effect from cannabis than those with ulcerative colitis. In a review of 20 studies in a 2020 issue of the Journal of Clinical Gastroenterology, researchers examined cannabis use among patients with IBD and found that cannabinoids had no effect on inflammatory biomarkers, and they were not effective at inducing remission, which is the ideal end point. However, patients who used cannabinoids reported significant improvements in abdominal pain, nausea, diarrhea, appetite and overall well-being. Similarly, a double-blind, randomized, placebo-controlled study in a 2021 issue of PLoS One found that patients with mild to moderate ulcerative colitis who smoked marijuana cigarettes daily for eight weeks—while continuing to take their usual UC medications—experienced improvements in their symptoms and quality of life, compared with those who were given placebo cigarettes, which contained cannabis flowers from which THC had been extracted. However, neither group experienced reduced inflammation, based on blood tests. All that said, it’s possible that the impacts of cannabinoids on symptoms could have trickle-down effects that decrease the need for other prescription drugs. For example, a study in a 2019 issue of the European Journal of Gastroenterology & Hepatology found that when patients with IBD used medical cannabis to treat their symptoms, their need for other medications was significantly reduced over the course of a year because their symptoms improved. Potential drawbacks A cautionary note: there’s a tipping point with using cannabis for GI disorders. “Cannabinoids reduce the tone of the lower esophageal sphincter, which can increase heartburn and reflux symptoms,” Kinnucan says. “They also decrease gut motility, causing the stomach to empty more slowly, which can increase nausea and be problematic for patients with gastroparesis,” a disorder that delays the movement of food from the stomach to the small intestine. Another potential risk: chronic, daily cannabis use can cause cannabinoid hyperemesis syndrome, which is characterized by recurrent nausea, vomiting and abdominal pain, Andrews notes. “Some people have many months with cannabinoid hyperemesis. Even if they stop using cannabis, it’s possible [their usage] may have induced a permanent change.” In addition, some develop a cannabis-use disorder, a form of dependence that occurs when the brain adapts to ongoing use of the drug. A study in a 2020 issue of the journal Drug and Alcohol Review found that approximately 27% of lifetime marijuana users develop a cannabis-use disorder, which is defined as problematic or continued use despite experiencing loss of control, social or medical problems, cravings, tolerance or withdrawal. “We don’t know what the right dose is where patients can have the positive effects and avoid the negative effects—and not all patients respond the same way to the same dose,” Kinnucan says. Plus, cannabis is used in many different ways—as edibles, smoking or vaping, dabbing, oils or tinctures—and the dosing is different with each route. “There’s little regulation of cannabis, and the THC levels are extremely high now,” Andrews says. Thirty years ago, the percentage of THC in typically accessible marijuana was in the single digits, he says, whereas these days THC is often 20% or higher. With these higher concentrations, “we have no idea what they will do to the [body’s] cannabinoid system long term,” Andrews says. There’s also a concern that people with IBD and other gastrointestinal disorders might stop using other treatments that have been approved by the FDA for their condition. “Because they feel better, they may have a false sense that they are better,” Kinnucan says. “It’s important to continue medical therapy to prevent progression of the disease. We know that medication non-adherence is associated with clinical relapse of IBD and could have implications on future disease outcomes.” Looking ahead, “what we need is to really start doing large, multicenter, randomized, controlled studies to examine the effects on IBD, using specific forms of cannabis at specific doses,” Tishler says. Until more is known, the onus is on patients to take precautions. For one thing, if you’re interested in trying it, find out what the laws are in your area: while some states have fully legalized marijuana, others allow it only for medical purposes, and still others continue to treat it as fully illegal. You’ll also want to find out what your employer’s policy is regarding medical marijuana use, in case there’s a chance that you may be drug-tested. “With chronic use, marijuana stays in your system for a long time,” Vaughn says. If you’re already using marijuana, whether for medical or recreational reasons, it’s important to tell your doctors—regardless of whether it’s legalized where you call home. Wherever you live, “you need to talk to your doctor about whether this is right for you,” Vaughn says. “It’s good to be open—your doctor is not going to be judgmental.” While this may seem like a privacy issue, it’s important to realize there could be medical risks. For one thing, cannabis can have potential interactions with other medications, such as warfarin (an anti-coagulant), benzodiazepines and barbiturates, Kinnucan warns. Cannabis use is also more likely to cause problems with certain groups of people, like those who are pregnant or breastfeeding, who have significant psychiatric disorders or who have a history of substance abuse, Poppers says. Finally, remember that experts primarily view cannabis as adjunctive therapy—a potential addition on an as-needed basis—for GI disorders. “This is not a panacea or a miracle drug,” Vaughn says. “For some people, it helps their symptoms, and for some people it doesn’t.” While Joe Silverman found that medical marijuana does help ease his Crohn’s disease symptoms, he has prioritized finding the most effective drug to treat the underlying cause of his condition. At the beginning of 2021, he and his doctor shifted the course of his treatment, and he began getting intravenous infusions of a different immunosuppressant drug every six weeks. “It has kept the inflammation [of my Crohn’s disease] at bay,” he says. Silverman continues to use medical marijuana for flare-ups or tougher days in a measured fashion. “With being able to measure these cannabinoid products by a dosed milligram each time, I still feel in control mentally and physically while reducing pain in my gut.” That’s the best of both treatment avenues. from https://ift.tt/3u6N9Q7 Check out https://takiaisfobia.blogspot.com/ |
Authorhttps://takiaisfobia.blogspot.com/ Archives
April 2023
Categories |
 RSS Feed
RSS Feed
