|
When a COVID-19 vaccine becomes widely available to Americans, Brianna Clarke says she won’t be taking it. “I don’t trust the vaccine,” the 22-year-old says. “I think it’s too soon to have a vaccine.” Clarke, who lives in Willingboro, N.J., is among a significant number of Black Americans who are skeptical of the healthcare industry in general and, critically, the COVID-19 vaccines recently developed by pharmaceutical companies Pfizer-BioNTech and Moderna, more specifically. Over the past couple of weeks, the U.S. has averaged in the neighborhood of 200,000 new COVID-19 cases per day, and about 2,500 related deaths, according to data compiled by John Hopkins University. Still, many Americans like Clarke are reluctant to get vaccinated. The rollout of the first authorized coronavirus vaccine, from Pfizer-BioNTech, began on Dec. 14, marking a critical moment in the U.S.’s battle against rising COVID-19 cases. Distribution of Moderna’s vaccine, authorized for emergency use on Dec. 19, began the following week. But for these vaccines to truly turn the tide of the pandemic, there will need to be near universal willingness among Americans to get the shot. There’s still uncertainty about exactly what number of Americans need to be vaccinated to reach so-called “herd immunity”—the level at which the virus will no longer be able to spread through the population—but Dr. Anthony Fauci, the director of the National Institute of Allergy and Infectious Diseases, predicted at a recent Harvard event that the number will need to be somewhere between 75 to 85%. Meanwhile, there’s a real concern that the distrust public health officials face with Black Americans could impact the country’s ability to reach that goal. According to a late-August/early-September poll by the Kaiser Family Foundation and The Undefeated, some 70% of Black Americans believe that people are treated unfairly based on race or ethnicity when they seek medical care. It’s a feeling born of unequal access to care and intensified by the pandemic, which is disproportionately ravaging Black lives both physically and economically. Since May, the rate at which the COVID-19 vaccine development process—what the Trump Administration dubbed “Operation Warp Speed”—is taking place has triggered fear and paranoia, particularly among Black Americans, that the ultimate product could be unsafe. Operation Warp Speed has been charged with producing and delivering 300 million doses of a COVID-19 vaccine by January 2021. The circumstances of the pandemic necessitate a quicker than usual development process, which has fostered misperceptions from many Black Americans that corners are being cut. Having a normally yearlong process of developing and distributing a vaccine telescoped into a matter of months—along with the name Operation Warp Speed—doesn’t instill confidence in many Americans, according Dr. Shirin Mazumder, an infectious disease specialist at Methodist LeBonheur in Memphis, T.N. “In the vaccine development trials, usually a vaccine takes about 15 to 20 years to develop, that’s what’s normal,” says Mazumder. Because of how quickly the COVID-19 vaccines have been developed, she points out “we don’t have long term data. Moderna and Pfizer have short term data, it’s only a couple of months…the long-term data is pending.” That might be one reason that Americans of color have been less likely to enroll in clinical trials for these vaccines, and others still in development. Of the 350,000 people who’ve registered online for a coronavirus clinical trial, 10% are Black or Latino, according to Dr. Jim Kublin, executive director of operations for the COVID-19 Prevention Network. That’s less than a third of the total U.S. population these two groups account for: 31.9%, according to U.S. Census data from 2019. Further, Centers for Disease Control and Prevention (CDC) data show that more than half of U.S. COVID-19 cases have been among Black and Latino people. “Like most of the other trials…the white participation just outpaces BIPOC communities. We need to be very proactive and mindful about ensuring that enough spots are allocated to POC so by the time trials are over, we don’t see the vast majority of the spots allocated going to white counterparts only,” says Dr. Michele Andrasik, who works at the COVID-19 Prevention Network, a government pandemic-response group housed in the National Institutes of Health. “It’s about ensuring that we are being transparent, providing accurate information in a timely manner, and we as researchers, as well as a scientific community, are showing that we are trustworthy.” A poll released by the Kaiser Family Foundation on Dec.15 revealed that among racial and ethnic groups, Black Americans are the most hesitant to get a vaccine, and continue to remain skeptical. In the survey, only 35% of Black adults said they definitely or probably would not get vaccinated, and were more likely than other groups to cite concerns about side effects and the newness of the vaccine. That tracks with the results of a Pew Research Center poll published earlier this month, in which only 42% of Black Americans said they would consider taking the vaccine, compared to 63% of Hispanic and 61% of white adults who would. These surveys come as the nation’s top health leaders urge Black people to trust the vaccine, hosting live events with Black health professionals at the forefront. In the focus group conducted in early October by Kaiser and The Undefeated, Black participants cited systemic racism for their vaccine hesitancy, noting the infamous, government-backed Tuskegee Syphilis Study. The U.S. Public Health Service Study at Tuskegee began in 1932 with the goal of tracking the damage the disease does to the human body. Without informed consent, the study enrolled 600 Black men, including 399 who had syphilis, according to the CDC. The participants were tricked into believing they were receiving free medical care but were instead just observed for a study of untreated syphilis. The men were told they were being treated for “bad blood,” and did not receive any treatment for their illness—not even after penicillin was found to cure syphilis in the mid-1940s. Dozens died as a result. The study did not end until it was exposed to the press in 1972 and has gone down as one of the nation’s most egregious examples of medical racism. But medical racism does not end there. Researchers have documented many ways that Black patients are treated differently from white patients in the U.S. For example, in its 2018 annual report, the Agency for Healthcare Research and Quality, part of the U.S. Department Health and Human Services, found that 40% of the measures of healthcare quality the agency assessed were worse for Black people than whites. Since the era of American slavery, Black people have faced mistreatment from many medical institutions. Like the Tuskegee Syphilis Study, Henrietta Lacks, who died in 1951 of cervical cancer, reflects the profiteering of Black bodies in the name of the advancement of science. Lacks entered John Hopkins Hospital seeking medical attention—and not receiving the same standard of care as white Americans. During an exam, her white doctor sampled her cells without her consent. For years, her own family had no knowledge that Lack’s cells were still alive in scientists’ labs, until they eventually discovered that scientists had used those original samples to fuel a cell line called “HeLa cells,” which has generated billions of dollars in pharmaceutical research and development. “My great grandmother and my family is one of the most well-known examples of what happens to African Americans in a medical industry, in reference to how we feel about our mistrust issues,” says Veronica Robinson, Lack’s great-granddaughter. “Because they launched a multi-million-dollar industry, which took 20 years for my family to even learn of, we have never been compensated for it.” These examples of malpractice are far from forgotten by the Black community. And all the while, the community has suffered incalculable damage due to ongoing and reprehensible health care disparities—which have only been exacerbated by the current pandemic. A November study by the American Heart Association showed that Black and Hispanic people have made up nearly 60% of COVID-19 hospitalizations in the U.S; a disproportionate number that researchers attribute to societal structures reinforcing health disparities among racial and ethnic groups: disparities in socioeconomic status, lack of access to health care, and to Black Americans being more likely to work essential jobs that increase the likelihood of exposure. Freeman A. Hrabowski III, the president of the University of Maryland at Baltimore County (UMBC), and his wife, Jacqueline, want to change attitudes about the vaccine in the Black community—and save lives. The Hrabowskis, who are Black, participated in a Phase 3 clinical trial for the Moderna Vaccine, conducted by the University of Maryland School of Medicine. “I am from Alabama,” says Hrabowski. “All my life, I knew about what was done to Black men and how they were abused, and the awful tragedy of the Tuskegee experiment. So, I can understand people questioning the motives of people when they talk about the study.” “But we must believe in the science, in spite of the challenge of our history and the way we’ve been treated. The reason I say that is, what choice do we have? Disproportionately large numbers of Blacks are contracting the disease and are dying. It helps for people to know that there have been people of different backgrounds, different racial backgrounds, in the development of the vaccine.” 
As of Dec. 21, participants in ongoing Pfizer-BioNTech vaccine studies are 10% Black or African American, 13% Hispanic, and 4% Asian, according to a company spokesperson. That’s actually relatively representative of the U.S. population, at least compared to the usual make up of drug studies, says Andrasik, of the COVID-19 Prevention Network. “They are not perfect, but they are closer to representing the censuses than the actual disparate impact of COVID.” While the medical industry insists it is moving with deliberate speed toward diversity and inclusion in experimental drug trials, some say prioritizing racial minorities is unlawful and unethical. “The issues with distributing the vaccine and who should get the vaccine first are really controversial and contentious,” says Jrvais Chen, a social epidemiologist at the Harvard T.H. Chan School of Public Health. “There has been some…debate on whether we should be targeting vaccines by race/ethnicity,” “On one hand, communities of color have been disproportionately affected,” says Chen. “On the other hand, there’s a lot of discomfort and lack of trust.” With a limited supply of shots, the deadly virus has turned allocation decisions into an ethical dilemma. There are ongoing concerns about the legal objections to any framework that uses race and ethnicity when determining vaccine allocation and the way it might be interpreted in court. For instance, strict judicial scrutiny would not permit health care workers to skip white people waiting in line for a vaccine in favor of a person of color, as Lawerence Gostin, a professor of global health law at Georgetown University, wrote in October in the Journal of the American Medical Association. Gostin also noted that public health agencies can not legally distribute vaccines to geographic locations based solely on race. Across all clinical trials, the National Academies of Sciences, Engineering, and Medicine (NASEM) says geographic priority should be given to communities that are high on the CDC’s Social Vulnerability Index, which identifies communities most endangered and in need of support during or after disasters. The index utilizes Census data that takes into account various socioeconomic, demographic, and housing vulnerabilities. “I dont think it’s about prioritizing one community over the other; it’s about ensuring inclusivity and ensuring equity across the trials,” says Andrasik. A CDC advisory group voted Dec. 20 that front-line essential workers will be next to recieve COVID-19 vaccines, in part to address racial disparities exposed by the pandemic. States and local governments have tried different strategies to contain the pandemic for months, including shutting down businesses and having people work from home. But a recent report from the Urban Institute indicates such strategies have little protection for communities hit hardest by the virus and who disproportionately hold jobs that must be done in-person. Researchers found that Black, Hispanic, and Indigenous workers are more likely than white workers to have jobs that must be done in-person and in close contact to other employees, putting them at an increased risk of contracting coronavirus. But allocating vaccines for these at-risk communities doesn’t solve the need to overcome the mistrust of the health care system that for some is deeply rooted. Dr. Reginald Eadie, president and CEO of Trinity Health of New England, says outreach to the Black community will be critical to get increased buy-in to the vaccine. “Otherwise, we would have a community that has unanswered questions and address concerns; a narrative that is directing their decisions and they would, in my opinion, be making a decision that’s not based on the facts,” says Eadie. 
Eadie has had frequent conversations with Black American community leaders to explain how the development process works and to address fears of receiving a vaccine that’s being rolled out in record time. “There were more than 200 people who attended our last conversation. We did a survey prior to our presentation, asking people how likely they are to take [the vaccine], and 38% of the audience said yes right away.” But after the conversation, he found that many of those who had said no had changed their mind. That, Eadie says, suggests a path forward: “You need to create ambassadors, then that vaccine hesitancy that communities of color are concerned about will begin to mitigate or disappear.” from https://ift.tt/2M2ZGSL Check out https://takiaisfobia.blogspot.com/
0 Comments
New York State Probes Fraudulently Obtained COVID-19 Vaccines Criminal Investigation Opened12/28/2020 (Bloomberg) -- A health care provider in New York may have fraudulently obtained COVID-19 vaccines and diverted them to members of the public in violation of priority guidelines, the state government said on Saturday. New York state police has started a criminal investigation into the matter, which could be the state’s first vaccine distribution-related scandal. The State Department of Health is aware of reports that ParCare Community Health Network, a provider with a clinic in upstate Orange County, “may have fraudulently obtained Covid-19 vaccine, transferred it to facilities in other parts of the state in violation of state guidelines, and diverted it to members of the public,” the state’s health commissioner Howard Zucker said in a statement. ParCare’s website shows it has six locations, including four in Brooklyn, New York, and says its patients are “mainly Orthodox Hasidic Jews, Hispanics, and African Americans.” A hyperlocal Orthodox Jewish news site, BoroPark 24, published a story on Dec. 21, saying ParCare had received a shipment of Moderna Inc.’s vaccines, and showed boxes of the vaccines in what appeared to be a refrigerator. It also had what appeared to be a copy of a ParCare advertisement touting availability of “The Newest Healthcare Revolution!” on a “first come first serve” basis. Gary Schlesinger, chief exeuctive officer of ParCare, was cited saying people who are a “health care worker, are over 60, or have underlying conditions” can register online to get a vaccine. That differs from state guidelines, which allow only frontline health care workers, or staff and residents of nursing homes, to get the first batch of vaccines. “We take this very seriously and DOH will be assisting State Police in a criminal investigation into this matter,” said Zucker in the statement. “Anyone found to have knowingly participated in this scheme will be held accountable to the fullest extent of the law.” Representatives with ParCare didn’t immediately respond to a request for comment sent on Saturday. from https://ift.tt/2WMStsl Check out https://takiaisfobia.blogspot.com/ A version of this article appeared in this week’s It’s Not Just You newsletter. SUBSCRIBE HERE to have It’s Not Just You delivered to your inbox every Sunday. ? Well hello! I’m so glad you’re here. This week I’ve got some thoughts on resolutions, ideas for finding winter joy, and the story of a waitress who survived COVID with a little help and is now paying it forward. And of course a comfort dog. BREAKING UP WITH NETFLIXIt’s the time of the year when we’re normally compelled to make outlandishly ambitious New Year’s resolutions. I’d argue that staying sentient up until this point feels like an accomplishment under these circumstances, and we could probably take it easy on the self-improvement quest next year. But you know we’re already plotting your reset. I only have one resolution, and it’s an embarrassing one. My goal in 2021 is to break up with Netflix–our relationship has gotten way out of hand. So here goes: Dear Netflix, I didn’t know how much you were going to mean to me when the pandemic began. I hadn’t looked at your homepage for months, but in March I watched Ozark. And after that, I felt like you finally got me. Your algorithm understood that I was the only one in the country not soothed by The Great British Baking Show or Gilmore Girls. You knew that in a national emergency what I really needed was a Black Mirror or Killing Eve. By June, I was visiting you every night, hungry for another paranormal procedural, a genre I didn’t know existed till you served it up. I tried to bargain with myself, vowing to spend time with you only when I was on my new pandemic exercise bike. Those were the days when we thought stay-at-home orders were also a once-in-lifetime chance to get your act together, develop good habits, stop with all your disordered disorder. I was on that bike for almost all eight seasons of Dexter, the show about a police blood-spatter analyst who is secretly a serial killer who kills serial killers. It was the ultimate in cardio multitasking. For a while I thought:
We learned so much about each other—or rather, you learned about me. You knew to suggest shows about Australians who come back from the dead in perfect health, as well as The Crown. What the common thread between those, but your algorithm did. By June, work video meetings had multiplied to the point that my Zoom fatigue was becoming Zoom paranoia. And the country started to crack from the strain of the pandemic and a long-overdue racial reckoning. I was too tired and sad to get on the bike. All I wanted was to curl up with you and the comforting justice of a vintage Law & Order: SVU episode, or the twisted morality of the old Twilight Zone. I’d find myself retreating to my bedroom earlier and earlier to spend time with you, and avoid everything else. <strong>I’d wake up at 2 a.m. and you’d be there asking: “Are you still watching?</strong>”Late at night, I’d whisper just one episode to the screen. One would turn into two or three. And then And instead of going back to sleep, I’d slip into a haze of rewinding, dozing, waking, and rewinding over and over as I tried to watch the parts of an episode I’d slept through. Each time missing the crucial scene that explained why the whole cast was suddenly on a helicopter crying. <strong>I’m ashamed to think of all the productive things I could have done with the 400+ hours we spent together this year, Netflix.</strong>There were other signs that our relationship was becoming unhealthy. I don’t think the 36 hours I spent with young Hannibal Lecter did my soul good; I blame you for introducing us. I’m starting to believe you’re holding me back. You might be the only thing standing between my lumpy pandemic self and the incredibly organized and fit woman I was sure I’d be by now. This is why I told my browser to block you in September. I only lasted a few days: watching cable news on Hulu is grimmer than a Swedish vampire movie. And your constant notifications about a witty dysfunctional single mom or the hot French detective show were irresistible. I’d pledge to only watch on my bike, but after one episode, I was back on the sofa binging the other 20 episodes in the middle of the night. These days, I don’t even know who I am without the bittersweet anticipation of a limited series. Plus the dog and I are getting chunky. But let’s face it. I can’t quit you cold turkey. Not at the start of this horrible winter. I can’t even stop buying chocolate every time I leave the house. And besides, experts say incremental changes are better than dramatic resolutions. So let’s try and make this a healthier relationship. Maybe, instead of fueling my darkest tendencies with your “slow burn” and “ominous” suggestions, your algorithm could sneak in some cheerier shows about couples who are not closet Zombies, or running a local heroin syndicate? Maybe you could find a DIY show I don’t hate. Just don’t get sappy on me, Netflix. Remember, this whole thing started when you the first time you said: Because you watched Ozark. ? 
SHARE this newsletter here. And, if you’re new to It’s Not Just You, SUBSCRIBE here. COPING KIT ⛱️? 6 Ways to Find Joy During This Dreary Winter From the CEO of the Happiness Research Institute in Copenhagen. ⛑️Why You Shouldn’t Make New Year’s Resolutions according to experts who say that setting big goals might be counterproductive this year. You see, I want a lot.</strong><br /> <strong>Perhaps I want everything</strong><br /> <strong>the darkness that comes with every infinite fall</strong><br /> <strong>and the shivering blaze of every step up.</strong> ? And, a word from Ludacris: 

<strong> I am a human being, meant to be in perpetual becoming. If I am living bravely, my entire life will become a million deaths and rebirths. </strong> Another Way to To Think About Your New Year’s Resolution Lots of us will be making our lists of things we want to change, or be, or achieve in 2021. For some hard-won life wisdom on aspiration, read Glennon Doyle’s acclaimed memoir Untamed. How are you feeling this winter? Send your thoughts and suggestions to me at [email protected] EVIDENCE OF HUMAN KINDNESS ❤️Here’s a small reminder of why building a network of support and person-to-person connection elevates everyone. This story is courtesy of Shelly Tygielski, founder of Pandemic of Love, a mutual aid community that matches those who want to give or volunteer directly with those who’ve asked for help with essential needs. 
Jennifer, a single mother, and waitress from Asheville, North Carolina, registered to get help from Pandemic of Love back in July when she was diagnosed with COVID-19 and sent home for several weeks. She explains: <strong>I rely on tips to get by and although it was still slower than usual due to the pandemic, I was managing to squeak by — then I got sick and everything started to collapse. I didn’t know how I was going to feed my kids</strong>“ Suzi Israel, the Asheville Pandemic of Love chapter leader stepped in and helped match Jennifer with a donor for groceries, utility bills, and past-due rent. After she recuperated and tested negative for the virus, Jennifer returned to work and has since become Pandemic of Love’s best ambassador. Last week, while waiting on a local couple, she told them all about her experience with the organization and they asked if she could connect them to Suzi. The pair, so touched by Jennifer’s passion and story, later called Suzi and arranged to meet her the following day, providing her with $1500 worth of supermarket gift cards for distribution to other local families with food insecurity so that they could have meals for the holidays. The chain of kindness never ends – it just keeps getting paid forward. Find out more about how to give or receive help from Pandemic of Love here. COMFORT DOG ?Our weekly acknowledgment of the creatures that help us make it through the storm. Meet TEDI the schnoodle shared by STEVE. Send comfort animal photos, suggestions, or comments to [email protected] 
? Did someone forward you this newsletter? SUBSCRIBE to It’s Not Just You here. from https://ift.tt/3aLAHh2 Check out https://takiaisfobia.blogspot.com/ (ATLANTA) — The United States will require airline passengers from Britain to get a negative COVID-19 test before their flight, the Centers for Disease Control and Prevention announced late Thursday. The U.S. is the latest country to announce new travel restrictions because of a new variant of the coronavirus that is spreading in Britain and elsewhere. Airline passengers from the United Kingdom will need to get negative COVID-19 tests within three days of their trip and provide the results to the airline, the CDC said in a statement. The agency said the order will be signed Friday and go into effect on Monday. “If a passenger chooses not to take a test, the airline must deny boarding to the passenger,” the CDC said in its statement. The agency said because of travel restrictions in place since March, air travel to the U.S. from the U.K. is already down by 90%. Last weekend, Britain’s Prime Minister Boris Johnson said the new variant of the coronavirus seemed to spread more easily than earlier ones and was moving rapidly through England. But Johnson stressed “there’s no evidence to suggest it is more lethal or causes more severe illness,” or that vaccines will be less effective against it. This week, New York Gov. Andrew Cuomo said three airlines with flights from London to New York — British Airways, Delta and Virgin Atlantic — had agreed to require passengers to take a COVID-19 test before getting on the plane. United Airlines on Thursday agreed to do the same for its flights to Newark, New Jersey. Britain has been under considerable pressure since the word of the new variant of the virus was made public. Some 40 countries imposed travel bans on Britain, leaving the island nation increasingly isolated. France relaxed its coronavirus-related ban on trucks from Britain on Tuesday after a two-day standoff that had stranded thousands of drivers and raised fears of Christmastime food shortages in the U.K. French authorities said delivery drivers could enter by ferry or tunnel provided they showed proof of a negative test for the virus. But the French restrictions were particularly worrisome, given that Britain relies heavily on its cross-Channel commercial links to the continent for food this time of year. from https://ift.tt/3hlKc7Z Check out https://takiaisfobia.blogspot.com/ (WASHINGTON) — Pfizer and BioNTech will supply the U.S. with an additional 100 million doses of the COVID-19 vaccine under a new agreement. The drugmakers said Wednesday that they expect to deliver all the doses by July 31. Pfizer already has a contract to supply the government with 100 million doses of its vaccine. Pfizer’s vaccine was the first to gain approval from the Food and Drug Administration and initial shipments went to states last week. It has now been joined by a vaccine from Moderna, which was developed in closer cooperation with scientists from the National Institutes of Health. from https://ift.tt/3plyr4u Check out https://takiaisfobia.blogspot.com/ Bill Gates isn’t about to be contemplating his touchdown dance yet. You don’t get to devote your life to battling the world’s most intractable problems by declaring victory before a fight is over. But in a letter Gates released this morning, headlined, “These breakthroughs will make 2021 better than 2020,” it’s clear that the co-director of the Bill and Melinda Gates Foundation is feeling pretty sanguine about the year ahead—particularly when it comes to COVID-19. The Gates letter, which comes a month before the expected January release of Melinda Gates’s Annual Letter, sees a lot to be optimistic about in the recent course of the pandemic—as well as a number of things to remain wary about. Clearly, Gates takes heart in the recent emergency use authorization of both the Moderna and Pfizer-BioNTech vaccines, as well as by the straightforward and decidedly lower-tech truth that face masks and distancing remain exceedingly effective in slowing the spread of the virus as the vaccines are being rolled out. “When I think back on the pace of scientific advances in 2020, I am stunned,” he writes. “Humans have never made more progress on any disease in a year than the world did on COVID-19 this year.” The Gates Foundation itself can take some credit for that breakneck pace, having been funding research on the mRNA technology that underlies both the Moderna and Pfizer-BioNTech vaccines since 2014. Not only is the technique effective, it makes for an easier-to-produce vaccine, since the mRNA shot causes the body itself to produce the spike protein that elicits an antibody reaction, rather than taking the time and making the effort to manufacture it in a lab. But that doesn’t mean it will be easy to produce the 5 to 10 billion doses of vaccine Gates estimates the world will need to slam the brakes on the pandemic—assuming two doses are necessary for some vaccines and an estimated 70% of the population must be inoculated to stop transmission. Together, all of the vaccine companies on the planet produce only 6 billion doses of vaccines against multiple diseases each year, Gates says. One way to accelerate production is through what are known as second-source agreements, which the Gates Foundation has helped broker and bankroll. The agreements pair vaccine developers like Pfizer with downstream manufacturers, which crank out the drugs the upstream companies invent. Much in the way automobile companies—which excel at mass production—retooled during World War II to manufacture tanks and other military armaments, so have high-output drug manufacturers like the Serum Institute of India, the biggest vaccine producer in the world, partnered with AstraZeneca to mass-produce the vaccine the latter company is developing. “They’ve already begun production, so there will be doses available for low- and middle-income countries if [Astra-Zeneca’s] vaccine is approved for use,” Gates writes. “And our foundation took on some of the financial risk, so if it doesn’t get approved, Serum won’t have to take a full loss.” Distribution is a potential choke point, of course. It’s one thing to produce 10 billion vaccines in a handful of manufacturing plants, it’s another thing to get them into billions of arms. The Gates Foundation is already working with 16 pharmaceutical manufacturers to make sure the vaccines are distributed widely and equitably, in cooperation with national governments which must take over the job of distributing the vaccines once they’ve arrived in-country. Gates takes heart in the fact that some of 2020’s ostensible failures have in fact been critical successes. There are only a few drugs that have, to date, been proven effective at treating COVID-19, among them dexamethasone and monoclonal antibodies. But at the beginning of the pandemic, there were potentially thousands of such drugs. Finding the few winners among them was made immeasurably easier by a collaboration among the foundation, Mastercard and the Wellcome Trust to develop the Therapeutics Accelerator, taking advantage of scanning technology that already exists in the pharmaceutical industry to screen the thousands of candidate compounds at high speed. The vast majority failed. “That was disappointing, but it was a useful disappointment,” Gates writes. “It spared the medical field millions of dollars and a year or two of laboriously going from one company to another, testing one compound after another.” COVID-19 surveillance is becoming easier too, thanks to the development of at-home tests that speed diagnosis and spare the public the painful nasal swabs that have become one of the signature indignities of the pandemic. Also in development are cell-phone sized devices that the foundation has helped deploy in 55 countries, able test a sample from a patient and automatically upload a positive or negative result—without the patient’s identity, in order to protect privacy—to a central database, speeding oversight of where the disease is spreading. Finally, Gates takes heart that most of sub-Saharan Africa—which is often harder hit than any other part of the world when a pandemic rages—is actually escaping with a comparatively lower case and mortality rate. Part of that is that the African population is young compared to most non-African nations, and young people tend to suffer less-severe cases of the disease. Part too is that in the continent’s extensive rural communities there is less time spent indoors, with families and crowds having less occasion to breathe the same air. Clearly, none of this means that the pandemic is beaten—or even that victory is close. But it does mean we have entered the end game. When the pandemic first began, Gates wrote in his blog, “this is like a world war, except in this case, we’re all on the same side.” That could have been too rosy a take. When human beings are pushed to our limits as the pandemic has done, we don’t always behave at our best. But researchers, manufacturers and the public have mostly stepped up to the challenge. And this world war will, before too long, looks like it will be won.
from https://ift.tt/3nJumGP Check out https://takiaisfobia.blogspot.com/ Welcome to COVID Questions, TIME’s advice column. We’re trying to make living through the pandemic a little easier, with expert-backed answers to your toughest coronavirus-related dilemmas. While we can’t and don’t offer medical or legal advice—those questions should go to your doctor or lawyer—we hope this column will help you sort through this stressful and confusing time. Got a question? Write to us at [email protected]. Today, Robert from Virginia asks:
Let’s start with the good news: Dr. Iahn Gonsenhauser, chief quality and patient safety officer at the Ohio State University Wexner Medical Center, agrees that your work environment sounds fairly low-risk. “A limited number of interactions that are brief—less than 15 minutes—with people who are wearing masks is pretty safe,” Gonsenhauser says. Still, if you choose to keep coming in, there are a few extra precautions you could request, Gonsenhauser says. Your supervisor should ask the building manager to implement capacity limits on elevators and mandate that everyone in the building (not just your suite) wear masks at all times. You could also ask for a “sneeze guard” in front of your desk and designate a specific package and document drop-off zone at least six feet away from your work area. And if you’re able, it’s safest to walk, bike or drive alone to work, rather than taking shared transportation. Even with COVID-19 precautions in place, though, “your fears are absolutely understandable and reasonable,” Gonsenhauser says. “Don’t keep how you’re feeling to yourself.” Your bosses sound pretty receptive to your concerns, which is good. It’s worth telling them that you simply don’t feel comfortable returning to work and would prefer to work from home. But here’s the bad news: There aren’t a ton of legal protections available to you if your bosses push back. Congress did pass a federal act that protects workers if they need to take leave to quarantine, care for someone who is quarantined or care for children whose school or daycare is closed—but none of that helps you. In fact, as you’ve seen, this policy “unfortunately makes those without children a prime target for bearing the burden of in-person work,” says Stacy Hawkins, an employment law expert at Rutgers Law School. The Occupational Safety and Health Administration (OSHA) also has pandemic safety recommendations for U.S. workplaces, but they’re non-binding. Fourteen states, including Virginia, have implemented COVID-19 workplace safety protocols on top of OSHA’s, which may provide stronger legal protections. The National Employment Law Project has a nice list of those here. Even still, office workers typically have the lowest level of legal protection, since their jobs are mostly considered low-risk, says Rachel McFarland, a staff attorney at the Virginia-based Legal Aid Justice Center. If your office isn’t following the guidelines set by OSHA and Virginia, your first step is bringing that to your bosses’ attention. In a perfect world, they’d make the necessary changes voluntarily—but if not, you could file an anonymous complaint with the state. (Just make sure you file in writing, McFarland says, because a phone complaint won’t trigger a site inspection.) But if your workplace is doing everything it should be doing and you simply don’t feel comfortable—which is perfectly fair!—there aren’t a ton of legal options available to you. McFarland says your best bet is to explain to your boss that you feel unsafe, and to point out that OSHA’s COVID-19 policy encourages employers to use teleworking and virtual meetings where possible. “I’d query how many emergencies there are that require somebody to be in the office,” she says. “It certainly seems that it’s possible to telework.” If they don’t agree, maybe there’s a compromise you could reach. Could you ask to work from home most days and stop by the office a few times a week? Are any of your coworkers willing to rotate in-person shifts so you don’t have to go in every day? If you do choose to resign, that’s understandable—but keep in mind that you may not be eligible for unemployment benefits if you voluntarily leave your job, Hawkins says. This is not an easy decision, and you’re not alone in having to make it. Good luck and stay safe. from https://ift.tt/3h9WErg Check out https://takiaisfobia.blogspot.com/ In recent weeks, the daily U.S. COVID-19 case and death counts have been hitting all-time highs, while fresh lockdowns and restrictions have been enacted across the country in an attempt to contain the spread of the virus. In response to the economic pain brought on by those measures, Congress is expected to pass a $900 billion economic relief package on Dec. 21. The relief package is a slimmed down version of the CARES Act, passed in late March, which included enhanced unemployment benefits, direct stimulus checks, and funds for schools and health care facilities. The new package is expected to provide a $600 direct stimulus payment to qualifying Americans and a $300 weekly supplemental jobless benefit. It will also expand the Paycheck Protection Program (PPP) for small businesses and provide relief for renters facing eviction. Before this most recent legislation, the government had authorized $2.3 trillion from the CARES Act and about $700 billion from various other economic aid packages for a total of just over $3 trillion dedicated to pandemic relief. Now, with the new measures, which arrived after months of congressional gridlock and last-minute negotiations, the government’s total fiscal response since the start of the pandemic stands at nearly $4 trillion. That’s a hard number to contextualize, but for comparison, it’s roughly equal to the entire combined annual economic output of Brazil, Australia and Mexico. It’s also comparable to the 2009 Recovery Act, an economic rescue package passed in response to the Great Recession—if that bill had been passed four times and adjusted for inflation, that is. But as a percent of GDP, the U.S. relief strategy still hasn’t been as aggressive as that of some other countries, according to a team of economists at universities in New York, South Korea and Turkey, who are following 168 nations’ fiscal and monetary responses to the pandemic. Their analysis shows that U.S. fiscal relief spending prior to the latest package was about 14% of GDP. The new package is likely to bump that ratio up, to roughly 18% of GDP. But that still puts the country behind other economically advanced economies like Japan (42% of GDP), Slovenia (25%) and Germany (20%). While the above chart shows how much financial weight each country has thrown behind its relief measures, it can’t show how effective that relief has been in stabilizing each economy. In the U.S., for example, the national poverty rate has risen faster than ever, due in part to lapses in economic relief after certain CARES Act provisions expired. Many European countries, meanwhile, had existing safety nets set up well before the pandemic that helped to cushion the blow for many affected residents. “Just the size of the package is never enough,” says Ceyhun Elgin, lecturer at Columbia University and professor of economics at Bogazici University in Turkey and one of the aforementioned researchers. “The content is what matters. There’s large variation in terms of how countries responded in the labor market. In some countries, there is support for parents whose kids are not in school. Others have direct food support, or compensation for sick workers.” In crafting each country’s specific relief provisions, global policymakers had to respond to their nation’s unique economic vulnerabilities; existing income and health safety-net systems; and standing in the global credit markets, among other factors. In the U.S., which has a unique capability to carry large amounts of debt, many provisions of the relief funding came in the form of direct payments, which the public will not repay. PPP loans, for instance, do not need to be repaid if firms use the funds to cover employee wages. In Europe, relief measures focused more on expanding existing furlough systems and loans to businesses, which could avoid driving up those countries’ national debts, assuming the loans are repaid. “Disposable incomes in the U.S. surged after direct checks were sent to households,” says Alejandra Grindal, a senior international economist at investment strategy firm Ned Davis Research, who has studied the global fiscal policy responses to the pandemic. “Europe didn’t see that kind of a jump.” Despite the staggering size of COVID-19 relief in the U.S., millions of Americans continue to struggle to cover basic expenses. In part, that’s because those funds have been so widely distributed—many Americans who did not lose income received direct payments alongside those who did, and businesses that were unaffected by the pandemic could still get PPP loans. While the U.S. relief efforts have been more like birdshot than a precision strike, funds have gotten some help to everyone relatively quickly and easily, political disputes notwithstanding. “Ideally we want to target those most impacted,” says Grindal. “But sometimes it’s easier to do blunt force.” from https://ift.tt/3mD0WbV Check out https://takiaisfobia.blogspot.com/ European Union Greenlights BioNTech-Pfizer COVID-19 Vaccine After Agency Gives Safety Approval12/21/2020 (AMSTERDAM) — The European Union on Monday gave official approval for the coronavirus vaccine developed by BioNTech and Pfizer to be used across the 27-nation bloc, raising hopes that countries can begin administering the first shots to their citizens shortly after Christmas. The EU’s executive commission gave the green light just hours after the European Medicines Agency said the vaccine meets safety and quality standards. Brussels had been expected to take two or three days to endorse the market authorization move. “As we have promised, this vaccine will be available for all EU countries at the same time, on the same conditions,” said European Commission President Ursula von der Leyen. “This is a very good way to end this difficult year, and to finally start turning the page on COVID-19.” Deliveries of the vaccine had been penciled in to start this coming Saturday, with inoculations beginning across the EU between Dec. 27-29, she said. The same vaccine was authorized in Britain and the United States weeks ago, prompting pressure from EU governments for EMA to speed up its approval process as virus cases surged again across the continent. EMA originally set Dec. 29 as the date for evaluating the vaccine, but moved up the meeting to Monday after calls from the German government and others for the agency to move more quickly. Harald Enzmann, the head of EMA’s expert committee, dismissed any suggestion that political influence had affected the decision. “The focus was exclusively on the science,” he told reporters. “That was a scientific assessment, full stop.” The Amsterdam-based EMA is responsible for approving all new drugs and vaccines across the 27 EU member states, Iceland, Liechtenstein and Norway. It is roughly equivalent to the U.S. Food and Drug Administration. The vaccine has already been given some form of regulatory authorization in at least 15 countries. Britain, Canada and the U.S. authorized the vaccine to be used according to emergency provisions, meaning the shot is an unlicensed product whose temporary use is justified by the pandemic that has killed almost 1.7 million people worldwide, according to a tally by Johns Hopkins University. Switzerland became the first country Saturday to authorize the vaccine according to the normal licensing procedure. EMA’s conditional market authorization also followed the regular process, only on an accelerated schedule and under the condition that the pharmaceutical companies submit follow-up data on their vaccine for the next year. While many have clamored for the vaccine’s authorization, there have also been concerns, in Europe and elsewhere, about the speed with which the shot was developed. Normally, vaccines take years to develop and approve, not months. In a statement last week that appeared to address those concerns, the agency stressed that the vaccine would only be approved after a scientific assessment showed its overall benefits outweighed the risks. Scientists are still waiting for more long-term follow-up data to see how long immunity from the vaccine lasts and if there are any rare or serious side effects. Final testing of the vaccine is still ongoing, and more information on whether the shot works in children is needed. EMA experts also said that data on pregnant women is limited, and physicians should decide on a case-by-case basis. The vaccine is not made with the coronavirus itself, meaning there’s no chance anyone could catch it from the shots. Instead, the vaccine contains a piece of genetic code that trains the immune system to recognize the spiked protein on the surface of the virus. On the day Britain began its vaccination campaign, authorities warned people with severe allergies not to get the shot after two people suffered serious allergic reactions; it’s unclear if the reactions were caused by the vaccine. The U.S. Centers for Disease Control and Prevention said that as of Friday there had been six cases of severe allergic reaction in the U.S. out of more than a quarter-million shots of the BioNTech-Pfizer vaccine given, including in one person with a history of vaccination reactions. Adding to an already grisly toll were concerns over a newly identified strain that appears to spread more easily. But experts expressed confidence that the new vaccine would still be effective against it. “At the moment, we can say that there is no evidence to suggest that the vaccine will not work against the new variant of the coronavirus,” Emer Cooke, the head of the drug regulator, said after a closed-door meeting in which experts unanimously recommended the shot for people over 16. But she added that protective measures such as wearing masks, washing hands and social distancing would likely be needed for some time. “Vaccines alone will not be the silver bullet that will allow us to return to normal life,” said Cooke. “But the authorization of this first vaccine is definitely a major step in the right direction and an indication that 2021 can be brighter than 2020.” The EU has ordered 300 million doses of the BioNTech-Pfizer vaccine; two are needed for each person, meaning other shots will be required to immunize all of the bloc’s nearly 450 million inhabitants. The EMA plans to hold a meeting on Jan. 6 to decide if another vaccine made by Moderna should be licensed. It has reviews ongoing for a shot developed by Oxford University and AstraZeneca and another from Janssen, but neither of those have made a formal request for the EMA to approve their vaccine. “Today is a particularly personal and emotional day for us at BioNTech,” said Ugur Sahin, the Germany-based company’s chief executive and co-founder. “Being in the heart of the EU, we are thrilled to be one step closer to potentially delivering the first vaccine in Europe to help combat this devastating pandemic.” ___ Jordans reported from Berlin. Associated Press writers Maria Cheng in Toronto and Lorne Cook in Brussels contributed to this report. from https://ift.tt/3avyExJ Check out https://takiaisfobia.blogspot.com/ A New More Contagious COVID-19 Strain Has Been Reported in the U.K. Is It Headed for the U.S.?12/21/2020 A new strain of COVID-19 reported in the United Kingdom has been blamed for a sharp uptick of cases—prompting new lockdowns in London and more than 40 countries to ban cross-border travel from the U.K. Although scientists say there is no evidence that the new strain is more deadly, Prime Minister Boris Johnson said it could be up to 70% more transmissible than others, while the health secretary said it was “getting out of control.” At least 40 countries including the entire European Union and Canada have temporarily banned incoming travel from the U.K. as of Monday. And the new strain has been detected in Denmark, Australia and Gibraltar, according to the British government; and in Italy and the Netherlands, according to media reports. But the U.S. has so far not stopped incoming travelers from the U.K. or Europe—sparking fears it may have already crossed the Atlantic Ocean. “Today that variant is getting on a plane and landing at JFK,” said New York Gov. Andrew Cuomo on Dec. 20, referring to New York’s busiest airport. “How many times in life do you have to make the same mistake before you learn?” Here’s what to know about the new strain of the virus. What do we know about the new strain of COVID-19?The strain was first detected by scientists in early December, according to the U.K. Health Secretary Matt Hancock. He first announced it on Dec. 14, saying that it was prominent in areas where the virus was spreading faster than expected. Retrospective analysis found the strain was first present in the U.K. as early as September, according to the government. By Dec. 19, scientific advisers to the U.K. government said they had “moderate confidence” that the variant was more transmissible than others, based on several factors including the exponential increase in infections despite lockdown measures. Genomic data suggests transmissibility that is some 71% “higher than other variants,” said a summary released by the U.K. government advisers. Researchers now believe a mutation to the genes that code for COVID-19’s spike protein, the part of the virus that clings to human cells allowing for infection, likely causes its increased transmissibility, according to a study published Dec. 18. However, what scientists know about mutation in SARS-CoV-2—the virus that causes COVID-19—is still evolving, as they collect more samples of the virus from cases around the world. The ongoing research means studies are leading to conflicting results about whether specific genetic changes are helping the virus to spread more easily, or cause more disease. In a Nov. 25 paper published in the journal Nature, for example, scientists studied more than 12,000 mutations of SARS-CoV-2 from viruses in 99 countries and concluded that none were more easily spreading from person to person. Read more: Inside the Global Quest to Trace the Origins of COVID-19—and Predict Where It Will Go Next Will vaccines still work against the COVID-19 variant?Officials said vaccines are still likely to work against the new variant, but that more research is being done to confirm this is the case. “The working assumption is that the vaccine response should be adequate for this virus, but we need to keep vigilant about this,” the U.K.’s chief scientific adviser Patrick Vallance said. “There is still much we don’t know,” Johnson said. “While we are fairly certain the variant is transmitted more quickly, there is no evidence to suggest that it is more lethal or causes more severe illness. Equally there is no evidence to suggest the vaccine will be any less effective against the new variant.” Still, because the current leading vaccines rely to some extent on targeting the spike protein, this mutation could be the first step in the virus becoming resistant to the current vaccines. “This virus is potentially on a pathway for vaccine escape, it has taken the first couple of steps towards that,” Professor Ravindra Gupta, professor of clinical microbiology at the Cambridge Institute of Therapeutic Immunology and Infectious Disease, told the BBC. “If we let it add more mutations, then you start worrying.” The U.K. began administering the COVID-19 vaccine developed by Pfizer and BioNTech on Dec. 8, following regulatory approval. What’s happening with cases in the U.K.?Daily confirmed cases in the U.K. hit new records over the weekend. On Sunday, 36,000 people in the U.K. tested positive for COVID-19—the highest one-day total yet recorded, and double the daily figures from earlier in the week. The number of people in the hospital is rising, and at the highest level since April. 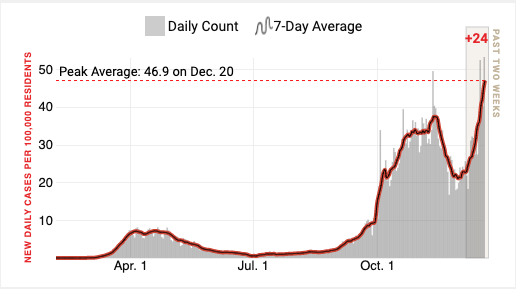
The spike comes despite several large swathes of the U.K. being under restrictive lockdown measures, with many businesses in some areas closed and people prevented from meeting indoors. Those measures looked to have caused a decline in daily new cases in late November. Government scientists say the new strain has driven the recent spike. Over the weekend, the U.K. government has placed London and the southeast of England into a new, restrictive “Tier 4” lockdown in response to the threat, forbidding people from traveling or meeting others over Christmas. The government had earlier said it would relax rules against groups meeting indoors over the Christmas period, despite calls from epidemiologists and opposition parties to think again. But as cases continued to rise in December, Johnson came under increasing pressure to change course, even as many families around the country planned to travel to see loved ones after a difficult year of isolation. As recently as Dec. 16, the Prime Minister had rejected calls for the rules to be tightened, accusing his opponents of wanting to cancel Christmas. (In the rest of the U.K., families will still be allowed to meet indoors, but only for Christmas Day instead of the five days over the festive period that had been allowed earlier.) Although experts say there is no evidence the mutation makes the virus any more or less deadly, an increase in people with the virus could result in hospitals becoming overwhelmed. “As numbers increase in the community, you will always have a proportion of people who will end up in hospital. If those numbers increase, the pressure on your hospitals increases because the background pressure on your hospitals is so high,” says Dr. Catherine Moore, a consultant clinical scientist at the Wales Specialist Virology Centre. That could lead to an increase in the death rate if there are shortages of trained medical staff available to treat them. “It doesn’t have to be more severe to be a problem,” Moore says. How has this impacted travel?At least 40 countries have now banned travel from the U.K. in light of the news about the new strain of the disease. But travel to the U.S. from both the U.K. and Europe is still allowed for American citizens, per Trump Administration rules in place since March. Right now, there are six flights per day into New York from the U.K., according to NBC New York. The U.K. newspaper The Telegraph cited aviation industry sources on Dec. 18 saying that the Trump Administration was planning to pass an executive order as early as Tuesday that would lift the ban on non-U.S. citizens arriving from the U.K. and Europe. The news from over the weekend may affect those plans. Within the U.K., there were scenes of chaos at train stations in London on Dec. 19 as people rushed to travel before new rules came into force. Many trains out of the capital were sold out, the Guardian reported. The government has now said people in London and the southeast of England, where cases are highest, must not meet others over the holidays. — With reporting by ALICE PARK/NEW YORK from https://ift.tt/3rjrti8 Check out https://takiaisfobia.blogspot.com/ |
Authorhttps://takiaisfobia.blogspot.com/ Archives
April 2023
Categories |
 RSS Feed
RSS Feed
