
This article is part of The D.C. Brief, TIME’s politics newsletter. Sign up here to get stories like this sent to your inbox. Across nearly every faith and in all but five states, a majority of Americans support at least some access to abortion. That’s a major conclusion of a new massive survey of 22,000 people from the Public Religion Research Institute. [time-brightcove not-tgx=”true”]But the survey’s most revealing insight is the group in which anti-abortion sentiment remains strongest: white Christian nationalists. On a state-by-state level, the more prone to Christian nationalism, the less likely PRRI found support for abortion. Statistically, it’s Mississippi at one end and Oregon at the other. Unsurprisingly, that spectrum also overlays neatly with the efforts to protect or scrap access to abortion. It’s a quiet chasm, but one that speaks to the latent threat of white Christian nationalism that Democrats and more than a few Republicans have been reluctant to confront head-on. “Clearly, the higher the score for Christian nationalism among state residents, there’s a clear linkage in terms of attitudes about abortion,” PRRI’s president Melissa Deckman told me this week ahead of the data’s public release. “It shows how clearly the Christian nationalist influences are making policy, and it’s really pretty stark.” Just 25% of those identified as Christian nationalists say abortion should be legal in all or most cases. White Christian nationalists were the most opposed to abortion, with a scant 29% weighing in on the side of abortion. Hispanic Christian nationalists (34%) and Black Christian nationalists (59%) were more supportive of abortion rights. Among those who completely rejected the tenets of Christian nationalism, the question of abortion rights is almost unanimously—93%—supported. The D.C. Brief has previously flagged the pernicious elements of Christian nationalism—specifically, white Christian nationalism—and its bleed into U.S. politics. The theology that the United States is a uniquely divined project that has its governance and godliness intertwined and blessed has been used aplenty by some of the most cynical political artists in the nation, and to great effect. A politician who can claim to be on a mission from God is not subject to the same level of fact-checking as one running on mortal rationality. In states with total bans on abortion, a staggering 53% of residents say they still support abortion rights in most or all cases. Put plainly: this aspect of democracy is not working, and it reveals a huge gap in our understanding of our neighbors. The threat here is not just to reproductive rights but also to the GOP brand for a generation. “I’m someone who studies Gen Z, and I’m telling you, Gen Z women are not buying anything that the GOP is selling these days. Part of it’s linked to their stance on abortion and LGBT rights,” Deckman says. Nationally, among all Americans, there is a whopping 50-point gap between Democrats and Republicans. A solid 86% of Democrats say abortion should be legal in all or most cases, while just 36% of Republicans say the same. To say the parties are working off different playbooks is an understatement. They’re not even working with the same set of rules. Abortion is shaping up to a defining issue in this campaign season. In a recent interview with TIME, former President Donald Trump basically confirmed a newfound nihilistic approach in which he pledged to leave it entirely to the states to decide their own levels of abortion access and regulation. But his strongest supporters are cheering on the crackdowns across red states. Florida, for instance, on Wednesday put into effect a ban on abortions at the six-week mark, ending its stand-alone status as an abortion haven in the South. The next closest state Southerners can turn to for the procedure is Virginia. Yet, as suggested in earlier surveys and confirmed yet again with PRRI’s data, only five states remain where supporters of abortion rights are in the minority: North Dakota and South Dakota at 47%; Arkansas at 46%; and Idaho and Utah logging in at 45% support. In the seven battleground states that are expected to define the presidential race—Arizona, Georgia, Michigan, Nevada, North Carolina, Pennsylvania, and Wisconsin—a collective 64% of residents say abortion should be legal in all or most cases. Even red states are seeing a whiff of goodwill toward abortion rights; 57% of residents there say abortion should be legal in all or most cases, including 59% of women. For decades, Democratic strategists treated abortion as a third-rail of an issue. Once Roe v Wade fell, that stopped being the case. Members of most religious traditions now see abortion as a part of the healthcare system that should be legal in most or all cases. Of the survey’s 16 breakout categories—including unaffiliated—just four have majority net disapproval of abortion rights. As expected, white evangelical Protestants top the roster with 72% saying abortion should be illegal in most or all cases. Latter-day Saints, or Mormons, follow at 69%, Hispanic Protestants say the same at 58%, and Jehovah’s Witnesses weigh in at 54%. But Jews, Catholics, mainline Protestants, and Muslims alike are all fine leaving the decision with patients and their health providers. That may explain why, when put to voters, protecting abortion rights has prevailed at the ballot box every time it’s been put there since the Dobbs decision. And it’s why, heading into the final push toward November, most Democrats have decided that abortion is not an inconvenient distraction best left on the shelf. White Christian nationalism and its loud minority may now be the albatross on U.S. policy making, but it’s a pretty useful demagogue, too. Make sense of what matters in Washington. Sign up for the D.C. Brief newsletter. from https://ift.tt/M8Ip3Fo Check out https://takiaisfobia.blogspot.com/
0 Comments

The first calls that Dr. Barb Petersen received in early March were from dairy owners worried about crows, pigeons and other birds dying on their Texas farms. Then came word that barn cats — half of them on one farm — had died suddenly. Within days, the Amarillo veterinarian was hearing about sick cows with unusual symptoms: high fevers, reluctance to eat and much less milk. Tests for typical illnesses came back negative. [time-brightcove not-tgx=”true”]Petersen, who monitors more than 40,000 cattle on a dozen farms in the Texas Panhandle, collected samples from cats and cows and sent them to Dr. Drew Magstadt, a friend from college who now works at the veterinary diagnostic laboratory at Iowa State University. The samples tested positive for a bird flu virus never before seen in cattle. It was the first proof that the bird flu, known as Type A H5N1, could infect cows. As of Wednesday, 36 U.S. herds had confirmed infections, according to the U.S. Agriculture Department. “It was just a surprise,” recalled Petersen. “It was just a little bit of disbelief.” At the same time, on almost every farm with sick animals, Petersen said she saw sick people, too. “We were actively checking on humans,” Petersen said. “I had people who never missed work, miss work.” So far, two people in the U.S. have been confirmed to be infected with H5N1, most recently a Texas dairy worker linked to the cattle outbreak, according to the U.S. Centers for Disease Control and Prevention. About two dozen people have been tested and about 100 people have been monitored since the virus appeared in cows, Dr. Demetre Daskalakis, a CDC respiratory diseases official, told reporters Wednesday. Daskalakis said CDC has seen no unusual flu trends in areas with infected cows, but some experts wonder if anecdotal reports of sick workers mean more than one person caught the virus from the animals. Petersen said some workers had symptoms consistent with flu: fever and body aches, stuffy nose or congestion. Some had conjunctivitis, the eye inflammation detected in the Texas dairy worker diagnosed with bird flu. Dr. Gregory Gray, an infectious disease epidemiologist at the University of Texas Medical Branch in Galveston, has been taking samples from livestock and people on two Texas farms. On farms with confirmed cattle infections, there have also been reports of mild illnesses among the workers, he said. His research has been difficult. Many workers are reluctant to be tested. That may be because they have limited access to health care or fear divulging private health information. Without confirmation, no one knows if the sick workers were infected with the bird flu virus or something unrelated, Gray said. “They seem to be linked in time and space, so one would say it’s biologically plausible,” said Gray. Some of the workers who fell ill sought treatment and were offered oseltamivir, an antiviral drug sold under the brand name Tamiflu, Petersen said. Some farm workers who were exposed to infected animals or people were offered the medication, CDC spokesman Jason McDonald said. State health officials are responsible for evaluating and providing treatment, according to federal guidelines. Health officials in Texas provided Tamiflu to the person known to be infected with H5N1 and household members, plus two people on a second dairy farm who tested negative but were exposed to infected animals, said Chris Van Deusen, a spokesman for the Texas Department of State Health Services. He said he wasn’t sure if others had been offered the antiviral. Farmers have been hesitant to allow health officials onto their land, said Dr. Kay Russo, a Colorado veterinarian who consulted about the outbreak with Petersen. “This particular disease is looked at as a scarlet letter,” Russo said. “It has this stigma associated with it right now.” Russo called for wider testing of cattle, people and milk. “We do not know what we do not measure,” she said. “Unfortunately, the horse left the barn and took off a lot faster than we were able to mobilize.” Gray worries that a recent federal order requiring testing of all lactating dairy cows moving between states could hinder cooperation even further. All labs that conduct tests must report positive results to the Agriculture Department. But many farmers may simply decide against testing, hoping to outlast the outbreak, he said. The reluctance of workers and farmers to allow testing is “greatly hampering” understanding of how the virus spreads, how large the outbreak is now and how quickly it may grow, Gray said. “It’s a negative, very negative, effect,” he said. Petersen said she understands workers’ and farmers’ fears. She praised the farmers who had been willing to let her gather the first samples that confirmed the outbreak and reflected on what the results could mean. “You immediately think about the cows, the people that care for them and the families that have these farms,” she said. “You’re thinking about the big picture, long term. Your mind starts to go down that entire path of concern.” from https://ift.tt/Nx8slaI Check out https://takiaisfobia.blogspot.com/ 
(PHOENIX) — Democrats secured enough votes in the Arizona Senate on Wednesday to repeal a Civil War-era ban on abortions that the state’s highest court recently allowed to take effect. Voting wasn’t complete but the Senate had the 16 votes it needed to advance the bill. Fourteen Democrats in the Senate were joined by two Republican votes in favor of repealing the bill, which narrowly cleared the Arizona House last week and is expected to be signed by Democratic Gov. Katie Hobbs. [time-brightcove not-tgx=”true”]The near-total ban, which predates Arizona’s statehood, permits abortions only to save the patient’s life — and provides no exceptions for survivors of rape or incest. In a ruling last month, the Arizona Supreme Court suggested doctors could be prosecuted under the 1864 law, which says that anyone who assists in an abortion can be sentenced to two to five years in prison. If the repeal bill is signed, a 2022 statute banning the procedure after 15 weeks of pregnancy would become Arizona’s prevailing abortion law. Still, there would likely be a period when nearly all abortions would be outlawed, because the repeal won’t take effect until 90 days after the end of the legislative session, likely in June or July. Several senators spoke about their motivations for voting as numbers were tallied on the repeal bill. “This is a clear statement that the Legislature does not want the territorial ban to be enforceable,” said Democratic state Sen. Priya Sundareshan, who voted yes to repeal. Arizona state Attorney General Kris Mayes called the vote “a win for freedom in our state,” but expressed concern that without an emergency clause, Arizonans would still be subject to the near total-abortion ban for some time. “Rest assured, my office is exploring every option available to prevent this outrageous 160-year-old law from ever taking effect,” she said. There were numerous disruptions from people in Senate gallery, as Republican state Sen. Shawnna Bolick explained her vote in favor of repeal, joining with Democrats. GOP state Sen. Jake Hoffman denounced Republican colleagues for joining with Democratic colleagues, calling it an affront to his party’s principles. “It is disgusting that this is the state of the Republican Party today,” Hoffman said. Advocates on both sides of the abortion issue arrived outside the Arizona Senate on Wednesday to emphasize their views. They included people affiliated with Planned Parenthood and faith-based groups opposed to abortion. A school-age girl kneeled in prayer in front of a table holding a large statute of the Virgin Mary, while a man with a megaphone shouted at passersby to repent. “I am expecting it will be repealed, but I am praying it won’t be,” said Karen Frigon, who was handing out brochures from the Arizona Right to Life. Arizona is one of a handful of battleground states that will decide the next president. Former President Donald Trump, who has warned that the issue could lead to Republican losses, has avoided endorsing a national abortion ban but said he’s proud to have appointed the Supreme Court justices who allowed states to outlaw it. The law had been blocked since the U.S. Supreme Court’s 1973 Roe v. Wade decision guaranteed the constitutional right to an abortion nationwide. When Roe v. Wade was overturned in June 2022 though, then-Arizona Attorney General Mark Brnovich, a Republican, persuaded a state judge that the 1864 ban could again be enforced. Still, the law hasn’t actually been enforced while the case was making its way through the courts. Mayes, who succeeded Brnovich, urged the state’s high court against reviving the law. Planned Parenthood officials vowed to continue providing abortions for the short time they are still legal and said they will reinforce networks that help patients travel out of state to places like New Mexico and California to access abortion. Advocates are collecting signatures for a ballot measure allowing abortions until a fetus could survive outside the womb, typically around 24 weeks, with exceptions — to save the parent’s life, or to protect her physical or mental health. Republican lawmakers, in turn, are considering putting one or more competing abortion proposals on the November ballot. A leaked planning document outlined the approaches being considered by House Republicans, such as codifying existing abortion regulations, proposing a 14-week ban that would be “disguised as a 15-week law” because it would allow abortions until the beginning of the 15th week, and a measure that would prohibit abortions after six weeks of pregnancy, before many people know they’re pregnant. House Republicans have not yet publicly released any such proposed ballot measures. from https://ift.tt/B3fmHKZ Check out https://takiaisfobia.blogspot.com/ 
The food we eat impacts every aspect of our lives and our bodies: our hormones, brain chemistry, immune system, microbiome; the list goes on. As consumers, we deserve the right to easily understand our foods’ nutritional value in order to make informed decisions about what we consume and how that will impact our health and well being. This is especially important when it comes to ingredients that are detrimental when eaten in excess, such as sugar. As researchers in functional medicine, longevity, AI, and nutrition, as well as inventors of health-enhancing and life-saving solutions, we have dedicated our professional lives to improving the health and well-being of millions everywhere. And while we applaud the Food and Drug Administration (FDA) taking important strides to pass mandatory front-of-package labeling for packaged foods in the U.S., this is a change that cannot come soon enough. Everyone’s health depends on it. [time-brightcove not-tgx=”true”]TheFDA recommends adults consume no more than 50 grams of added sugar per day (based on a 2,000 calorie diet), but the average American consumes closer toone-third of a pound of sugar daily, more than three times the recommended amount. To put that into perspective, the average American consumesover 100 pounds of sugar per person per year. With that much sugar consumption, it is no wonder that49% of American adults are diabetic or pre-diabetic. What’s worse is that much of the sugar we consume occurs without our even realizing it. There are over 60 different ways sugar is identified on nutrition labels, making a consumer’s attempt to regulate their sugar intake unfairly complicated. Extensive academic research published in medical peer-reviewed journals backs common knowledge that excess sugar consumption can lead to serious chronic conditions, as well as fatigue, anxiety, memory loss, ADHD, and even to a shorter life. Seventy four percent of packaged foods in the U.S. contain added sugar, including seemingly healthy foods, such as salad dressing, coleslaw, and even baked beans, marinades, and yogurt; somesweetened yogurts contain more sugar than a can of soda. The fact that sugar is so biologically addictive — studies indicate it iseight times more addictive than cocaine — makes the reality that it’s hidden in so many foods even more harmful. Most of us are addicted to sugar and we don’t even know it. This cycle of addiction is relentless and hard to break: we eat food with sugar, which then triggers a blood sugar spike, which lights up the pleasure center in our brain. When the inevitable sugar crash comes, we seek that spike again in the form of craving more sugar. Without easily discernible food labeling, shoppers unknowingly create this cycle inside their own bodies, even while they erroneously think the food they’re buying is healthy. Read More: How the World Got Hooked on Sugar In many countries, labels on packaged foods serve a similar function to labels on cigarette cartons: to warn consumers of risk. InChile, a policy of “high in” labels on the front of sugary drinks dramatically reduced the consumption of those beverages. In Israel, afront-of-package labelling system, wherein a red label indicates an item high in sugar, has led to significant positive changes in 76% of the population’s food buying habits. We’re excited to see what a similar program in the U.S. would yield. Those in the U.S. lobbying against this front-of-package change, unsurprisingly, have an interest in the continued popularity of their products. In a February 2023joint filing, the nation’s largest cereal producers threatened a lawsuit after proposed changes would not allow them to label products as “healthy” if they didn’t meet nutritional standards. The front-of-package suggested change would rightfully prevent many cereals on the market with excess sugar from calling themselves “healthy.” This dynamic is similar to changes made in cigarette advertising in the 20th century. In the 1940s, a famous Camel cigarettes campaign featured theslogan, “More doctors smoke Camels.” By 1969, a mandatory warning label was added to cigarettes, giving consumers clearer access to information about risks, allowing them to make more informed choices about their health. Today the percentage of Americans who smoke is 11% compared to nearly50% back in the day when “more doctors smoked Camels”. Life expectancy rosenearly 11 years in that span of time too, and thedecrease in smoking certainly contributed. While front-of-package labeling on packaged foods is a crucial first step towards a healthier society, education and awareness alone will only get us so far. To drive even more significant change in the way most Americans eat, a change that will lead to a healthier population, we must also incentivize the production and widespread distribution of healthier alternatives. These alternatives—a packaged cookie with healthier ingredients, for instance—must be just as delicious, and readily available as those loaded with sugar. The recently announced new standards by the U.S. Department of Agriculture (USDA) that will limit added sugars in school meals can greatly help with the availability of healthier alternatives, especially when children form their eating habits. For the rest of us, though, front-of-package labeling is an important step one in this journey towards national wellness and it will also encourage producers to create healthier options for consumers; readily available healthier alternatives is step two. FDA leadership ensuring labeling of high contents of sugar in packaged foods could increase awareness and reduce the negative impacts of sugar and help millions live healthier longer lives. This change would help us make more informed choices about our food and our health. We believe it is our right, and every American’s right, to have clear and visible information about the sugar content of the foods we are eating in order to make more informed decisions. from https://ift.tt/CnfIdts Check out https://takiaisfobia.blogspot.com/ 
At least a dozen people in California and Washington have been sickened with E. coli food poisoning linked to organic walnuts sold in bulk in 19 states, U.S. health officials said Tuesday. The nuts were sold in natural food and co-op stores such as Whole Foods and Market of Choice. Seven people have been hospitalized and two have developed a dangerous kidney disease known as hemolytic uremic syndrome, officials with the Centers for Disease Control and Prevention said. Gibson Farms Inc. of Hollister, California, has recalled potentially affected walnuts with expiration dates between May 21, 2025 and June 7, 2025, the Food and Drug Administration said. Some stores may have repackaged bulk walnut pieces into clamshells or bags. The FDA has a list of stores where the walnuts were sold. The nuts are potentially contaminated with dangerous E. coli bacteria that can cause severe stomach cramps, diarrhea, including bloody diarrhea and vomiting. Symptoms start three to four days after consuming the food. Most people recover within five to seven days. Consumers who bought organic walnuts from bulk containers should check to see if they’re part of the recall. Recalled nuts should not be sold or served, the CDC said. Wash items and surfaces that may have come in contact with the nuts using hot soapy water or a dishwasher. Contact a health care provider about any symptoms. from https://ift.tt/Cdg916Z Check out https://takiaisfobia.blogspot.com/ 
Nearly 40% of LGBTQ+ youth have seriously considered attempting suicide in the last year, according to a survey by the Trevor Project, a national suicide prevention organization that operates a hotline and collects data on the mental health of queer youth. More than 18,500 LGBTQ+ people living in the U.S. between the ages of 13 and 24 participated in the survey, which was conducted between September and December 2023. [time-brightcove not-tgx=”true”]The findings are part of the organization’s sixth annual survey results, and have been released in a year where more than 480 anti-LGBTQ+ bills have been introduced across the country. Last year, attempted rollbacks against LGBTQ+ rights were even greater, with the American Civil Liberties Union tracking more than 500 similar state bills. These policies have a serious effect on youth: 90% of survey respondents said their well-being was negatively affected by recent politics. And 45% of transgender and nonbinary youth even reported that they or their family have even considered moving to a different state because of local LGBTQ+ policy and laws, according to the 2024 Trevor Project survey. Read More: Crisis Center Receives 500% Increase in Calls Following Nex Benedict’s Death Dr. Ronita Nath, vice president of research at the Trevor Project, says the data helps create a snapshot of the current mental health landscape of LGBTQ+ youth. The survey is meant to supplement the Centers for Disease Control and Prevention’s (CDC) Youth Risk Behavior Survey for high school students in the U.S., which was released in February 2023. That data, which surveys a nationally representative sample of U.S. high school students, found that LGBTQ+ youth reported experiencing feelings of sadness or hopelessness at a rate of 69%, compared to heterosexual students at 35%. The 2024 Trevor Project survey shows the need for more welcoming communities. Queer youth who reported being physically threatened, harmed, bullied, discriminated against, or subject to conversion therapy had suicide attempt rates that were double than those who did not share those experiences. Most youth reported that these incidents happened to them in school, with 32% saying they were verbally harassed because others thought they were part of the LGBTQ+ community. Another 21% said that they were not allowed to dress in a way that fit their gender identity or expression. “LGBTQ plus people are not inherently prone to greater suicide risk because of their sexual orientation, gender identity, but that they aren’t placed at greater risk because of the stigma and discrimination they experience and in society,” Nath tells TIME. “We do urge politicians, members of the media, anyone publicly debating about LGBTQ+ young people to really understand that… protecting and supporting the mental health and well being of young people, you know, should not be up for political debate.” More From TIME
[video id=FPjceZSA autostart="viewable"]
Access to careThe 2024 Trevor Project survey comes shortly after the UCLA School of Law’s Williams Institute own survey that reveals that 93% of transgender youth aged 13 to 17 in the U.S. live in states that either have passed or are considering bills that would limit their access to gender-affirming-care, bathrooms, proper pronoun usage, or participation in sports that match their gender identity. While much political discourse focuses on medical care that could change one’s physical appearance, an overwhelming majority of LGBTQ+ youth, nearly 85%, reported wanting mental health care. Yet only 1 in 2 LGBTQ+ youth were actually able to receive that care. “Our research really illustrates how intangible barriers like stigma and fear play a really major role in preventing people from accessing mental health care,” says Nath. Young people cited concerns like being afraid to talk about their mental health with someone else, lack of affordability, and not wanting to have to get their caregiver’s permission as the biggest obstacles to receiving mental health care. “While 24 states have passed laws banning and criminalizing best practice and essential medical care for transgender nonbinary young people, our survey found that very few people received this care to begin with. Just 13% reported being on gender-affirming hormones, and 2% reported taking puberty blockers,” Nath says. Still, of those on hormones, 61% were somewhat or very concerned about losing access to that care. How to help LGBTQ+ youthSurvey respondents were asked to list ways people can best support LGBTQ+ youth. 88% of respondents wanted people to trust that they knew who they were. Four in five people said they wanted people to show up for them. 77%asked that others not support politicians working to advance anti-LGBTQ+ legislation. “We can encourage schools in our communities to implement, you know school district policies that prioritize suicide prevention,” says Nath. “We can advocate for intersectional approaches to mental health care for students. We need to have zero tolerance policies for anti-LGBTQ+ bullying and harassment.” If you need to talk to someone about your mental health or suicidal thoughts, please call The Trevor Project at 1-866-488-7386 or the National Suicide Prevention Lifeline at 1-800-273-8255 or 988. from https://ift.tt/hLM8NfF Check out https://takiaisfobia.blogspot.com/ 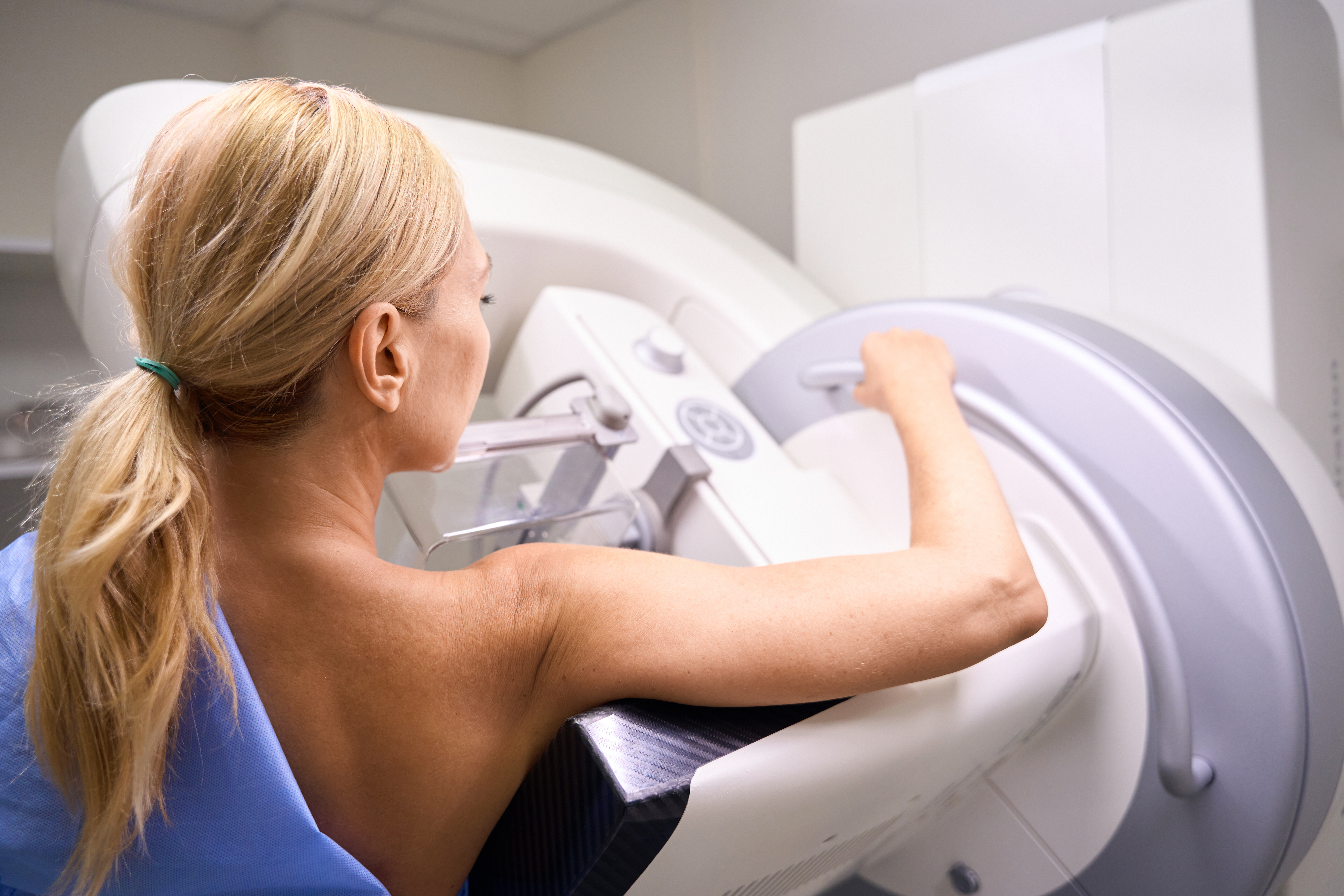
Most women should start mammogram screenings for breast cancer at age 40, and get screened every other year until they reach age 75, according to new recommendations from an expert panel. The U.S. Preventive Services Task Force (USPSTF), which is an independent group of experts funded by the government, regularly reviews data and makes recommendations on health issues, and many health providers follow them. It decided to revise its advice on mammogram screening that was last issued in 2016. That guideline said women should start regular mammogram screening every other year beginning at age 50, and that women ages 40 to 49 should discuss with their doctors the best screening regimen for them. [time-brightcove not-tgx=”true”]Here’s what to know about the latest change. When should most women get their first mammogram?The new recommendation is based on additional evidence that has emerged since 2016, says Dr. John Wong, vice chair of USPSTF. According to data from the National Cancer Institute, death rates from breast cancer for women in their 40s began increasing by 2% annually in 2015, and that trend justified a change in the recommendations to start screening a decade earlier. “Our current data shows that this recommendation could potentially save as many as one out of five women who would otherwise die if they waited to be screened until they were 50,” says Wong. “That’s potentially saving 25,000 women from dying of breast cancer. We think that’s a big win.” Read More: Why Are So Many Young People Getting Cancer? Dr. Maxine Jochelson, a radiologist at Memorial Sloan Kettering Cancer Center, says the revision is long overdue. “The data have shown for years that by not screening women between ages 40 and 50, if women in that age group develop breast cancer, they are more likely to need chemo, more likely to need larger surgery, and more often get more aggressive cancers,” she says. “I think they are late to the party.” It’s not clear what is contributing to increased risk among women in their 40s. But Wong says the task force analyzed whether USPSTF’s recommendation around that time—to start mammograms once women turned 50 rather than 40, as the group’s previous guideline advised—was a factor, as some advocates had warned. “Screening rates remained consistent throughout that period,” he says. “So that’s not the cause.” The most recent data do include different populations of women, however, incorporates different types of screening and treatment options that weren’t available when the previous populations were studied, so more screening may be leading to more diagnoses, for example. The current recommendation now brings the USPSTF’s guidance more in line with that of other health groups including the American Cancer Society. That group advises women to start screening at age 45 annually until age 54, then every other year. Why did the recommendation change?Wong says the new guidelines reflect the changing benefits and risks of screening and its consequences, which include additional testing, as well as the risk of false positives. The increased risk of dying from breast cancer among women in their 40s tipped the balance in favor of beginning screening earlier. What about women with dense breasts?About half of women in the U.S. have dense breast tissue; for them, mammograms are less reliable at detecting cancer. The task force is less clear about whether these women should follow the same recommendations. It says the evidence supporting the benefits of additional screening—with MRIs or ultrasounds, which doctors often recommend if mammograms are negative or inconclusive—isn’t “sufficient.” Wong says more research is needed to understand if those additional imaging tests help women to get diagnosed earlier and ultimately allow them to live longer. “We just don’t have clear evidence at this time,” he says. Will insurance cover mammograms starting at age 40?All insurance companies (with few exceptions) must cover the cost of mammograms with no co-pay for women who get them as part of regular screening beginning at age 40. That’s part of the Protecting Access to Lifesaving Screenings Act that was passed by Congress in 2019. Because of this act, the new guidelines should not affect insurance coverage of mammograms for women in their 40s. Read More: A5-Minute Quiz Revealed Olivia Munn’s Breast Cancer Risk. You Can Take It Too But since the task force says the evidence for additional screenings is “insufficient,” women with dense breast tissue may still have to pay out of pocket for additional tests beyond a mammogram. That could lead to lower follow-up for these women and ultimately may delay any breast cancer diagnoses until later stages, when the disease is harder to treat. “We worry about what it means for access and utilization for those women, to say that there is inconclusive evidence to support supplemental imaging,” says Molly Guthrie, vice president of policy and advocacy at the Susan G. Komen Breast Cancer Foundation. Guthrie notes that already, many states require that mammography centers notify women if they have dense breast tissue, so they and their doctors are aware that the mammogram readings may have missed potential red flags for cancer. That requirement will apply to all mammography facilities beginning this September, after the U.S. Food and Drug Administration, which regulates the facilities, issued a new rule last year mandating the information. “The FDA is trying to get to the point of pushing the conversation about dense breast tissue so women have a dialogue with their providers,” says Guthrie. “But if you are not doing anything to change coverage, you are not going to increase utilization.” Wong stands by the task force’s conclusion, seeing it as an invitation for further study. “We would love to have sufficient evidence that would help women with dense breast tissue to live longer, healthier lives, and we are urgently calling for more research to obtain that evidence,” he says. “We always look at the latest and best science—and at the benefits and harms—to make recommendations that help people in this nation stay healthy and live longer.” from https://ift.tt/7fSk2Fy Check out https://takiaisfobia.blogspot.com/ 
Joe Biden’s presidential campaign criticized Donald Trump on Tuesday for saying that, if elected, he would close an office in the White House tasked with making sure the country is better prepared for the next pandemic. In an interview with TIME published Tuesday, Trump said he would disband the Office of Pandemic Preparedness and Response Policy (OPPR), which opened last summer after Congress approved a bill in 2022 with bipartisan support to mandate its creation. The office most recently responded to an outbreak of bird flu in dairy farms, coordinating with the Food and Drug Administration to ensure milk remains safe to drink, and working with farmers to contain the virus. [time-brightcove not-tgx=”true”]Trump described the office to TIME as “a way of giving out pork” and said an effective pandemic response could be mobilized once a virus emerges. “I think it sounds good politically, but I think it’s a very expensive solution to something that won’t work. You have to move quickly when you see it happening,” Trump told TIME. The Biden campaign compared Trump’s comments to his haphazard response to the COVID-19 pandemic during his last year in office, when he claimed the virus would disappear “like a miracle” and would “go away without a vaccine” and suggested during a White House press briefing that the virus could be cured by injecting patients with bleach. “Pandemic preparedness isn’t abstract to the millions of Americans that lost a loved one because of Donald Trump’s failed response to COVID-19,” said Kevin Munoz, a senior campaign spokesman and the former spokesman for White House’s COVID-19 response during the beginning of the Biden administration. “We know the impact of Trump’s inability to lead all too well: an economy in shambles, schools closed down, and far too many American lives needlessly lost. We cannot afford to go back.”
In late 2022, Congress, seeking to add more resources to prepare for future pandemics, passed the PREVENT Pandemics Act, formally establishing the Office of Pandemic Preparedness and Response Policy. The office is currently run by retired Maj. Gen. Paul Friedrichs. The Biden White House has requested $6.2 million to fund the office in fiscal year 2025. The office keeps Americans ready for biological threats and pathogens, says Dr. Raj Panjabi, who previously served as Biden’s top NSC official for pandemic preparations. Shutting down the OPPR “would leave Americans, as it did in 2020, deeply unprepared to respond to a pandemic and run the risk of leading to the same kind of chaotic response that we all saw with bodies piling up in trucks that had to be converted into morgues in New York City and ultimately to a loss of lives that are preventable and avoidable,” he says. Trump’s comments, adds Panjabi, “just shows he’s not serious about it now, which is just absolutely absurd after over a million Americans lost their lives to this.” Trump also claimed in his interview with TIME that previous administrations had not prepared better for a pandemic than he did. “I’m not blaming the past administrations at all, because again, nobody saw it coming,” Trump said. “But the cupboards were bare. We had no gowns, we had no masks. We had no goggles, we had no medicines. We had no ventilators.” It’s true that when Trump came into office, the U.S. Strategic National Stockpile, an integrated collection of secret, federally-controlled warehouses packed with medical and protective equipment, was short on supplies. That’s because President Barack Obama deployed resources from the stockpile for public health emergencies such as the swine flu and the Ebola outbreak. When Obama tried to refill the stockpile, Tea Party Republicans blocked the new funding. In his first three years as President, Trump never attempted to replenish the equipment. That proved costly once the pandemic struck. By April of 2020, the U.S. government had already distributed 90 percent of its supplies. —WITH REPORTING BY ERIC CORTELLESSA from https://ift.tt/6h2BdDy Check out https://takiaisfobia.blogspot.com/ 
(WASHINGTON, D.C.) — The U.S. Drug Enforcement Administration will move to reclassify marijuana as a less dangerous drug, The Associated Press has learned, a historic shift to generations of American drug policy that could have wide ripple-effects across the country. The DEA’s proposal, which still must be reviewed by the White House Office of Management and Budget, would recognize the medical uses of cannabis and acknowledge it has less potential for abuse than some of the nation’s most dangerous drugs. However, it would not legalize marijuana outright for recreational use. [time-brightcove not-tgx=”true”]The agency’s move, confirmed to the AP on Tuesday by five people familiar with the matter who spoke on the condition of anonymity to discuss the sensitive regulatory review, clears the last significant regulatory hurdle before the agency’s biggest policy change in over 50 years can take effect. Once OMB signs off, the DEA will take public comment on the plan to move marijuana from its current classification as a Schedule I drug, alongside heroin and LSD. It moves pot to Schedule III, alongside ketamine and some anabolic steroids, following a recommendation from the federal Health and Human Services Department. After the public-comment period the agency would publish the final rule. It comes after President Joe Biden called for a review of federal marijuana law in October 2022 and has moved to pardon thousands of Americans convicted federally of simple possession of the drug. He has also called on governors and local leaders to take similar steps to erase marijuana convictions. “Criminal records for marijuana use and possession have imposed needless barriers to employment, housing, and educational opportunities,” Biden said in December. “Too many lives have been upended because of our failed approach to marijuana. It’s time that we right these wrongs.” The election year announcement could help Biden, a Democrat, boost flagging support, particularly among younger voters. Schedule III drugs are still controlled substances and subject to rules and regulations, and people who traffic in them without permission could still face federal criminal prosecution. Some critics argue the DEA shouldn’t change course on marijuana, saying rescheduling isn’t necessary and could lead to harmful side effects. On the other end of the spectrum, others argue say marijuana should be dropped from the controlled-substances list completely and instead regulated like alcohol. Federal drug policy has lagged behind many states in recent years, with 38 having already legalized medical marijuana and 24 legalizing its recreational use. That’s helped fuel fast growth in the marijuana industry, with an estimated worth of nearly $30 billion. Easing federal regulations could reduce the tax burden that can be 70% or more for businesses, according to industry groups. It could also make it easier to research marijuana, since it’s very difficult to conduct authorized clinical studies on Schedule I substances. The immediate effect of rescheduling on the nation’s criminal justice system would likely be more muted, since federal prosecutions for simple possession have been fairly rare in recent years. Biden has already pardoned thousands of Americans convicted of possessing marijuana under federal law. ___ Goodman reported from Miami, Mustian from New Orleans. AP writer Colleen Long contributed. from https://ift.tt/Rbq9gXQ Check out https://takiaisfobia.blogspot.com/ 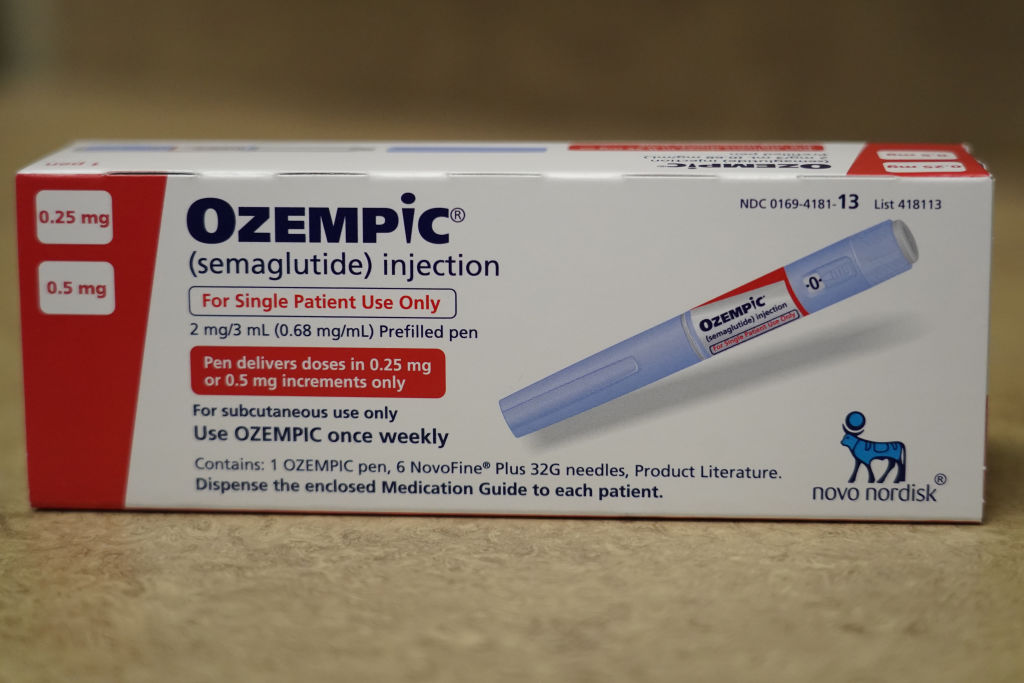
Nearly a decade ago, a thin, soft-spoken twenty-something woman named Megan walked into my office and presented me with a meticulous hand-written file containing her gastrointestinal history. The file included descriptions of her initial diagnosis of Crohn’s disease as a teenager, the multiple operations she had endured to remove diseased parts of her bowels, and the array of symptoms she suffered with, including nausea, a dozen bowel movements a day, and a total reliance on nutrition obtained through her veins, known as total parenteral nutrition (TPN). Instead of twenty feet of small intestines that most people have, she only had around twenty inches left, and was diagnosed with a condition called short bowel syndrome. We tried a variety of treatments in the ensuing months, but she remained miserable and mostly homebound, unable to concentrate on her studies or to take short vacations with her friends. Finally, I started her on a relatively new injectable drug called Gattex (teduglutide), an analogue of a human hormone secreted by intestinal cells known as glucagon-like peptide-2 (GLP-2). Gattex improved intestinal blood flow and absorption, slowing gastric emptying and increasing the height of the tiny, finger-like projections in Megan’s intestines known as villi. Six months or so after she started the drug, Megan was not only free of gastrointestinal symptoms: she was also able to discontinue her TPN on weekends, a freedom previously unimaginable. Gattex is an expensive drug, and appropriate for use only in certain clinical contexts for rare-disease patients. The drug was slow to garner attention within or outside of gastroenterology. In the years that followed, I continued to use it for patients like Megan. For those patients, it seemed nothing short of a miracle. [time-brightcove not-tgx=”true”]Today, another glucagon-like peptide, GLP-1, is widely hailed as miraculous. GLP-1, like GLP-2, is a hormone secreted by intestinal cells and is being studied in short bowel syndrome patients. Unlike Gattex, however, drugs that mimic GLP-1 (GLP 1s) are currently prescribed for some of the most common chronic conditions in the U.S. today—diabetes and obesity—and have been showered with media attention over the last couple of years in the context of weight management. Traditionally, GLP-1s were developed to treat type 2 diabetes. These injectable drugs resemble hormones that the body produces after eating, stimulating insulin secretion by the pancreas and lowering blood sugar levels. They slow digestion and decrease appetite. They affect parts of the brain that control hunger, telling your brain to feel full for a longer period of time. Not surprisingly, GLP-1s can lead to lower body fat—a welcome side effect for many diabetic patients struggling with weight issues. Some GLP-1s are now FDA approved for chronic weight management, including Wegovy (semaglutide) and Saxenda (liraglutide), while others like Ozempic (semaglutide) are approved for type 2 diabetes but are used off-label for weight management—including for cosmetic weight loss, after being popularized by celebrities and social media influencers. If GLP-1 agonists like Wegovy and Ozempic are modern wonder drugs, their effects on body weight and blood sugar are only a part of their sensational story. A lesser-known feature of GLP-1s—and GLP-2s–is their potential to lower local and systemic inflammation within the body, both in the intestines and beyond. We know today that inflammation can be an important risk factor for the development of all kinds of disease. Low-level inflammation is linked to a wide variety of chronic conditions, including heart disease, cancer, obesity, diabetes and neurodegenerative disorders, all of which can be considered, at least in part, chronic inflammatory disorders (CIDs). In obese individuals, excess body fat—particularly the visceral fat stored deep inside the body—churns out low-level inflammation at all hours of the day. Inflammation may be one central mechanism by which obese individuals develop additional CIDs, including top killers like heart disease and cancer. New and emerging research suggests that GLP-1s, acting through anti-inflammatory pathways, may be beneficial in a variety of CIDs, with the potential to aid not only patients with diabetes and obesity but also those without these conditions. Recent data reveals that GLP-1s are useful in heart disease, a condition that is currently the leading cause of death in men and women worldwide. In August of 2023, a study sponsored by Wegovy manufacturer Novo Nordisk was published in the New England Journal of Medicine (NEJM). Researchers tracked 529 patients with obesity and heart failure, assigning them to receive either weekly semaglutide or placebo for one year. They found that the patients treated with semaglutide had not only greater weight loss than those on placebo but also fewer symptoms and physical limitations as well as improved exercise tolerance. Read More: Why Ozempic Can’t Fix America’s Obesity Crisis A few months later, in November of 2023, results from a landmark clinical trial known as the SELECT trial, which was also sponsored by Novo Nordisk, caused a stir. Around 17,000 cardiovascular disease patients who were overweight or obese but did not have diabetes were randomized to receive weekly semaglutide or placebo for several months. At the end of the study period, researchers found that the patients who had received semaglutide not only lost weight: they had a stunning 20% decrease in cardiovascular events, including cardiovascular death, heart attacks, and strokes. In addition, semaglutide decreased heart failure and all-cause mortality by 18% and 19%, respectively. Interestingly, semaglutide seemed to be preventing heart attacks within the first couple of months of taking the drug, before study participants lost much weight, supporting the idea that patients didn’t need to lose weight before starting to experience the drug’s cardiovascular benefits. Those taking the highest dose of semaglutide in the SELECT trial had drops in systemic inflammation. This reduction in inflammation may be one important mechanism by which semaglutide yields its cardiac benefits. Studies have shown that elevated low-level inflammation is an independent risk factor for the development of heart attacks, strokes and death from cardiac events. Semaglutide also helped to improve traditional risk factors for heart disease, including overweight and obesity as well as elevated blood pressure, blood sugar and cholesterol levels. These traditional risk factors, in turn, are also linked to chronic, low-level inflammation. Beyond heart disease, GLP-1s may have a role in other CIDs. Over the last few decades, the incidence of early-onset cancer, meaning cancer diagnosed in adults less than 50 years of age, has been on the rise. This emerging global epidemic is thought to be due in large part to environmental and lifestyle factors like a suboptimal diet, lack of exercise and pollution—all of which can trigger inflammation in the body. In December of 2023, researchers at Case Western University published results from a nationwide observational study involving over one million patients with type 2 diabetes who were prescribed antidiabetic medications from 2005-2019. Compared with other antidiabetic medications, including insulin and metformin, GLP-1s were associated with a decreased risk for colorectal cancer, a finding that held fast regardless of whether the patient had diabetes alone or diabetes in addition to overweight or obesity. Observational studies cannot prove causation, and figuring out how GLP-1 impacts cancer will likely be complex given the multifactorial nature of cancer causation, which includes both genetic and environmental influences. Still, we know today that inflammation is one of the hallmarks of cancer. Inflammation, in many cases, fuels the initiation and development of cancer, from early genetic and epigenetic influences that transform normal cells into malignant ones to the continued growth and spread of cancer throughout the body. GLP-1s may influence the pathways that promote cancer not only by controlling weight and blood sugar but also through their anti-inflammatory effects independent of these traditional risk factors. A significant proportion of patients with diabetes or obesity are destined to develop kidney disease. Practitioners deploying GLP-1s for diabetes noticed that these drugs also seemed to stabilize patients’ kidney function. Both experimental and, more recently, clinical trials have shown that GLP-1s can slow kidney decline and prevent substantial loss of kidney function in diabetic patients. GLP-1s like semaglutide and liraglutide have been shown to protect the kidneys through alteration of fat and energy metabolism. Patients taking GLP-1s tend to have better blood sugar control, lower body weight, and lower blood pressure, all of which can also decrease inflammation and positively impact kidney function. In addition, researchers at Monash University in Australia published a study earlier this year revealing that the GLP-1 agonist liraglutide, with receptors in the kidneys, can suppress inflammation and decrease markers of kidney damage in diabetic and non-diabetic mouse models of kidney disease. Another common complication in patients with diabetes or obesity is metabolic dysfunction-associated steatotic liver disease (MASLD), previously termed non-alcoholic fatty liver disease (NAFLD). MASLD is now the most prevalent chronic liver disease in the Western world and affects around a third of the US population. Meanwhile, licensed pharmacotherapies for this disease are lacking. In MASLD, droplets of fat accumulate in the liver, which may in turn lead to liver inflammation and scarring over time—meanwhile, inflammation itself may help to feed this pathway. Weight loss is central to reversing early-stage disease. GLP-1s are currently being evaluated for the treatment of MASLD. Randomized controlled trials show that semaglutide can lower fat and inflammation in the liver and improve markers of liver injury, although it has not yet been shown to improve the stages of liver scarring. Semaglutide treatment is also associated with a decrease in body wide low-level inflammation in patients at risk for developing MASLD. Low-level inflammation is predictive of MASLD and has been linked to the presence and severity of underlying liver scarring. Trials are ongoing to determine if semaglutide will meet the clinical endpoints for FDA approval. Read More: Ozempic Hurts in the Fight Against Eating Disorders GLP-1s have received attention in lung and intestinal diseases. Results from a few clinical trials as well as observational studies have shed light on the potential benefits of GLP-1s in asthma, emphysema and chronic bronchitis, although large-scale clinical trials are required to confirm these findings. Obesity, which creates ongoing, chronic inflammation in the body, is increasingly associated with inflammatory bowel disease (IBD) and may negatively influence the disease course. Scientists also hypothesize that obesity contributes to the development of IBD. Some studies have shown that IBD patients taking GLP-1s for diabetes reduce the need for steroids or biologic medications and have fewer hospitalizations or IBD-related surgeries, suggesting that GLP-1s may influence disease activity in IBD. Further research is needed, however, to definitively determine whether GLP-1s are truly beneficial for disease outcomes in IBD patients. How might GLP-1s decrease inflammation in the body? Improved control of CIDs like diabetes and obesity lowers body fat and blood sugar levels, which means less inflammation. But GLP-1s have been shown to have anti-inflammatory effects independent of body weight or blood sugar. Human trials reveal that they reduce blood inflammatory biomarkers, especially when compared with other standard anti-diabetic treatments. And they seem to affect both local and body wide inflammation. GLP-1s have anti-inflammatory effects in various tissues, reducing the production of inflammatory cytokines and preventing the movement of immune cells into tissues. Receptors for GLP-1s are expressed on some immune cells–speaking to their potential to modulate the immune system and inflammation—as well as other cells around the body. They are found not only in the pancreas but in a wide variety of organs, including the heart, kidneys, lung, liver, blood vessels, stomach, intestines and brain. In fact, the prevalence of GLP-1 receptors in the body helped to prompt initial interest in studies investigating the use of GLP-1s for disorders other than diabetes. Yet the distribution of immune cells containing GLP-1 receptors in various tissues is highly unequal, suggesting that the actions of GLP-1s in some organs may be more important than in others when it comes to regulating inflammation. Immune cells activated by GLP-1s are especially replete in the intestines—which house much of the body’s immune system—as well as in the brain. According to Canadian scientist Dr. Daniel Drucker, whose pioneering research gave rise to modern GLP-1 drugs, the gut-brain-immune axis may be central to the ability of GLP-1s to influence inflammation in the body, as his latest study conducted in animals and published earlier this year suggests. The bidirectional communication between the gut and the brain incorporates signals from trillions of gut microbes collectively known as the intestinal microbiota and affects a plethora of processes around the body, including inflammation. GLP-1s may modulate gut microbiota and target immune cells in the brain and in the gut to reduce body wide inflammation. Given the potential benefits of GLP-1s in the brain, it’s no surprise that there is rising interest in the use of GLP-1s for neurodegenerative diseases. Controlled trials point to the ability of GLP-1s to reduce the rates of dementia in diabetic patients, and large-scale clinical trials are underway to study the effectiveness of these drugs for diseases like Alzheimer’s and Parkinson’s. GLP-1s are thought to modulate immune function in the brain, preventing the continued buildup of misfolded proteins and lowering inflammation—pathways that contribute to Alzheimer’s and other neurodegenerative diseases. For all the possibilities GLP-1s seem to have in managing inflammation and CIDs, caution must be exercised when evaluating and deploying these drugs outside of their traditional contexts. Their anti-inflammatory effects are broad, targeting different pathways in different tissues, and their range of potential benefits may be accompanied by problems as well. Although GLP-1s are generally well tolerated, with common side effects including gastrointestinal issues like nausea, vomiting, diarrhea and constipation, rare, more serious complications include issues like stomach paralysis, pancreatitis, bowel obstruction, kidney failure and thyroid cancer. Long-term safety data in non-diabetic populations is required for the newer GLP-1s that are more effective for weight loss. The drugs are also expensive, often requiring significant out-of-pocket costs. In short, GLP-1s shouldn’t be used as a quick-fix in order to lose a few pounds, but rather for patients in whom the risk-benefit profile makes sense—in obese individuals at a high risk of developing additional CIDs, for example, in whom the use of GLP-1s seems less concerning than the risks associated with a lifetime of severe weight issues. For many obese individuals, metabolic derangements, including an altered hormonal milieu, can make it tough to lose and keep the excess weight off indefinitely without adjunctive medical or surgical therapies. Unfortunately, the people who most need GLP-1s for weight management often struggle to obtain it. Prescribing GLP-1s for obesity—or any chronic disorder—doesn’t obviate the need for lifestyle changes, which are paramount to decreasing inflammation and the risk of developing CIDs. In fact, certain lifestyle changes, like incorporating more soluble fiber—a type of fiber that feeds our gut microbes--into the diet, or exercising regularly, can increase endogenous GLP-1 levels. Lifestyle prescriptions may also help to lessen the risks of GLP-1s by allowing patients to be maintained on the lowest effective dose, or to even wean off GLP-1s entirely at some point. GLP-1 drugs like Ozempic and Wegovy gained immense clinical traction as well as popular cultural fame due to their success in treating overweight and obesity, conspicuous diseases that burden over two-thirds of all American adults. However, their effects may be broader than ever imagined: evidence is accruing on the potential of GLP-1s to combat invisible, low-level inflammation as well as several of the chronic conditions associated with it. But while GLP-1s may hold promise for treating a variety of diseases, more research is needed to establish exactly which patient populations beyond existing ones may benefit from these drugs. In the meantime, patients should discuss the potential utility of GLP-1s for a particular condition with their physicians. And as in the case of my patient Megan, who walked into my office long ago with a dire rare disease, the use of glucagon-like peptides–which are powerful gut hormones–should not be cavalier. from https://ift.tt/OJnfM3u Check out https://takiaisfobia.blogspot.com/ |
Authorhttps://takiaisfobia.blogspot.com/ Archives
April 2023
Categories |
 RSS Feed
RSS Feed
